Patient-centered Care Approach
- Read more about Patient-centered Care Approach
- Log in to post comments
The severity of TB disease is crucial for management strategies and treatment outcomes.
The National TB Elimination Programme (NTEP) follows an integrated algorithm for screening and ruling out active Tuberculosis (TB) among Household Contacts (HHCs) of Drug-resistant Tuberculosis (DR-TB) patients.
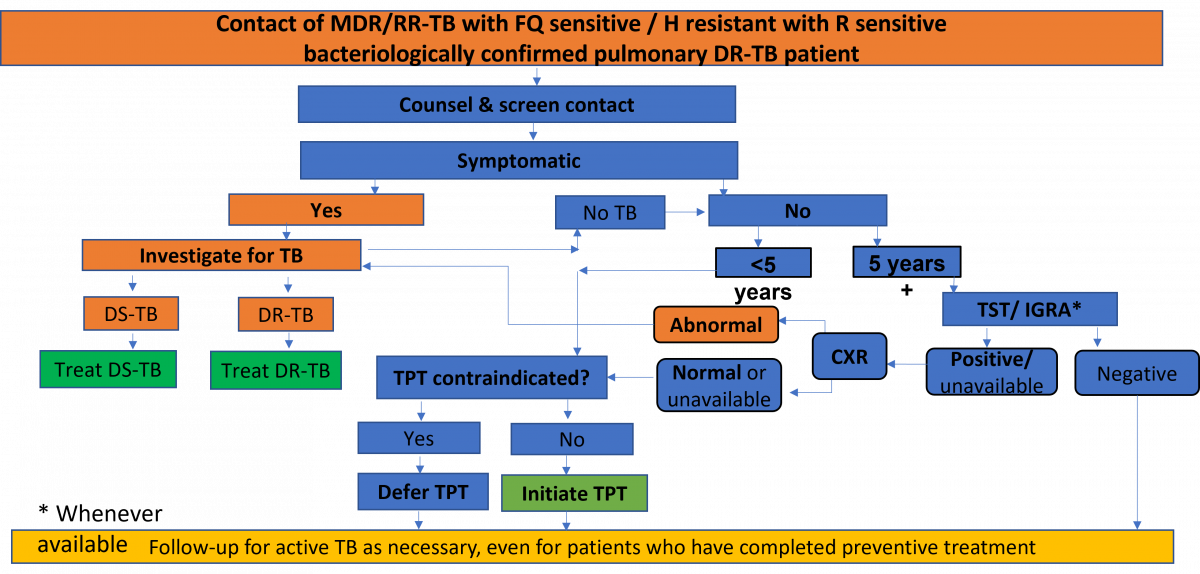
Figure: Integrated Algorithm for Screening and Ruling out Active TB among HHCs of DR-TB Patients; Source: Guidelines for PMDT in India, 2021, p118.
The suggested treatment regimen covering maximum non-Mycobacterium Tuberculosis (NTM) mainly Mycobacterium Avium Complex (MAC) is as follows:
Intensive Phase (IP) is for 3 months and can be extended to a maximum of 6 months with all four drugs.
It is very important that medical care continues and the patient is not abandoned once the therapy has been suspended. Several supportive measures can be used for this.
Dyspnoea, and to a much lesser extent pain, are among the most distressing symptoms in the last stages of the disease. Thus, access to opioids and other controlled medicines is fundamental for delivering high standards of palliative and end-of-life care to patients with Multidrug-resistant Tuberculosis (MDR-TB).
The details on palliative care supportive measures are as below:
The concept of Patient Turnaround Time (P-TAT) is to find out how much time was taken from the identification of the patient for a test to getting the result of that test and initiation of patient's treatment based on the test result.
The National TB Elimination Programme (NTEP) have set benchmarks to monitor the P-TAT as provided in the table below.