Management of Hepato-toxicity During Treatment with Shorter/Longer Oral MDR-TB Regimen
The management of hepatotoxicity during treatment with a shorter/ longer oral MDR-TB regimen is elaborated in the figure below. Careful monitoring of Alanine aminotransferase (ALT) and Aspartate aminotransferase (AST) levels is important to delineate the line of management.
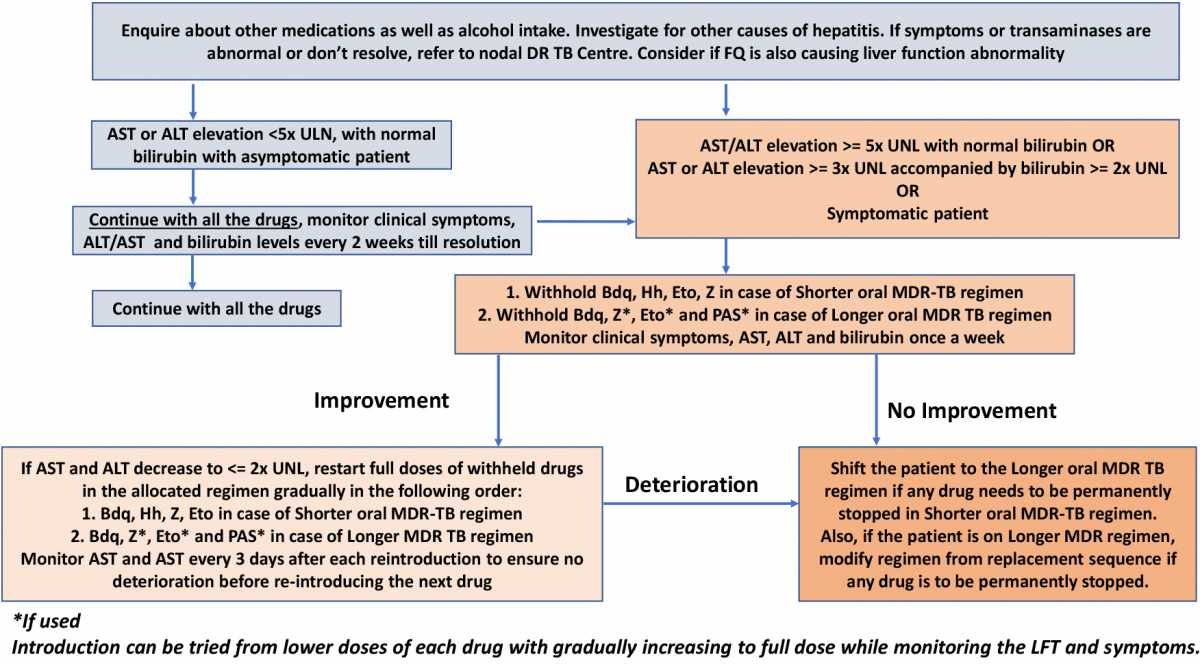
Figure: Management of Hepatotoxicity During Treatment with Shorter/ Longer Oral MDR-TB Regimen; Source: Guidelines for PMDT, India, 2021, p89.
Resources
Additional Monitoring During Pregnancy and Lactation in Women on Shorter/Longer Oral MDR-TB Regimen
Additional monitoring is recommended for pregnant women managed with a shorter/ longer oral Multidrug-resistant TB (MDR-TB) regimen including foetal and neonatal monitoring.
| INVESTIGATION | INDICATION / FREQUENCY |
|---|---|
| Basal Thyroid Stimulating Hormone (TSH) in Pre-treatment Evaluation (PTE) |
TSH Deranged in PTE - Monthly TSH Normal in PTE- Quarterly |
H Mono/Poly DR-TB Patient Flow from Diagnosis to Treatment Initiation
If Isoniazid (H) mono/ poly Drug-resistant TB (DR-TB) is detected by First-line Line Probe Assay (FL-LPA) then:
Distribution of Second-line Drugs from the District Drug Store to the TB Unit Store
The National TB Elimination Programme (NTEP) follows a standard procedure to distribute drugs to the Tuberculosis Units (TUs) in which the key role is played by District Drug Store (DDS) pharmacist.
For distribution of the drugs to the TUs, National Health Mission (NHM)/ NTEP allows for the hiring of the vehicle or utilizing the available vehicle.
Preparation of Patient-wise Boxes [PWB] for DR-TB Patients
Under the National TB Elimination Program (NTEP), Drug-resistant TB (DR-TB) patients receive standardized DR-TB treatment boxes (called Patient-wise Boxes - PWB) for each month. These boxes are either with the health facility nearest to DR-TB patients (DOT centre) or with the treatment supporter who is involved in the care of DR-TB patients.
Steps involved in the preparation of second-line drug boxes for DR-TB patients:
Treatment Algorithm for MDR/RR-TB
- Read more about Treatment Algorithm for MDR/RR-TB
- Log in to post comments
The treatment algorithm for Multidrug-resistant/ Rifampicin-resistant TB (MDR/ RR-TB) patients is a part of an integrated diagnostic and treatment algorithm under Programmatic Management of Drug-resistant Tuberculosis (PMDT).
DR-TB Treatment Care Cascade
- Read more about DR-TB Treatment Care Cascade
- Log in to post comments
The Drug-resistant TB (DR-TB) treatment process for all DR-TB patients follows a care cascade.