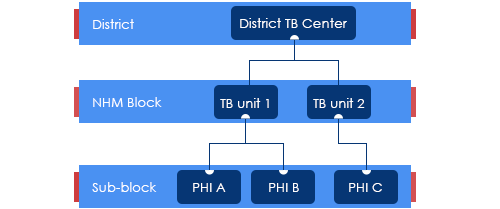
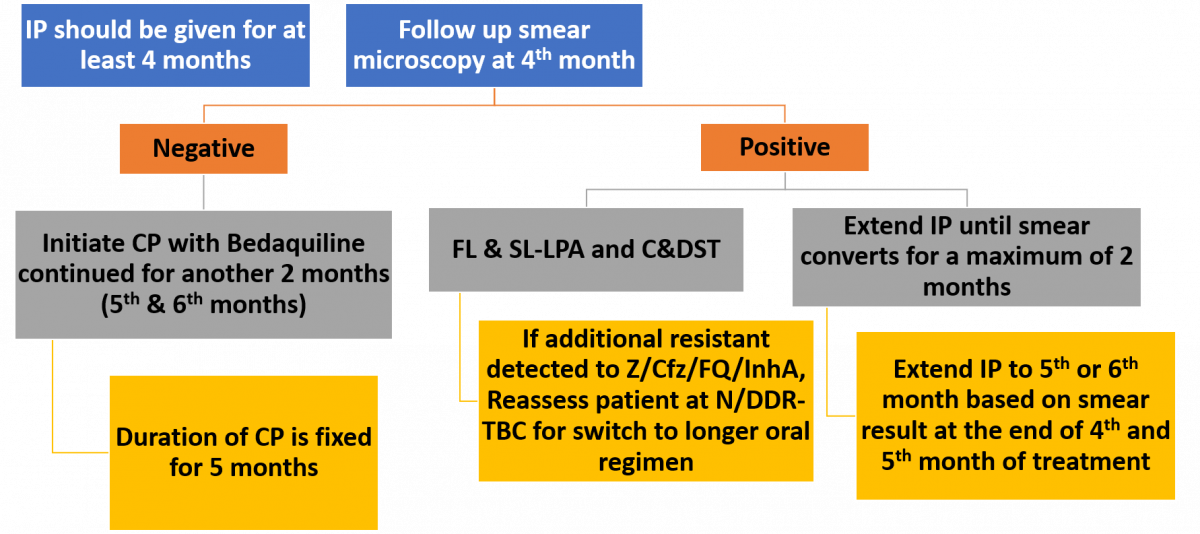
Treatment Regimen for Non-Mycobacterium Tuberculosis [NTM]
The suggested treatment regimen covering maximum non-Mycobacterium Tuberculosis (NTM) mainly Mycobacterium Avium Complex (MAC) is as follows:
- Rifampicin (R) 450-600 mg OD
- Ethambutol (E) 800-1200 mg OD
- Clarithromycin (Clr) 1 gm OD (split into two doses)
- Add injection Amikacin (Am) 750 mg – 1 gm thrice weekly for the first 2-3 months
Intensive Phase (IP) is for 3 months and can be extended to a maximum of 6 months with all four drugs.