-
PHI-Pharmacist: TB and TB Epidemiology
FullscreenTuberculosis
Content
Figure: Causative agent for Tuberculosis is Bacillus: Mycobacterium tuberculosis (M.tb)
-
Tuberculosis (TB) is a communicable disease that is a major cause of ill health.
-
TB is caused by the bacillus Mycobacterium tuberculosis (M.tb)
-
TB disease typically affects the lungs (pulmonary TB) (80%) but can also affect other parts of the body (extra pulmonary TB) (20%)
-
It spreads when people who are sick with TB expel bacteria into the air (for example by coughing, sneezing, shouting or singing)
-
It is one of the top 10 causes of death worldwide and the leading cause of death from a single infectious agent
Resources
- Global Tuberculosis Report, 2020; Geneva: World Health Organization, 2020
- Training Modules (1-4) for Programme Managers and Medical Officers India: Central TB Division, MoHFW, Government of India,July 2020
-
Global Burden of TB
Content- Globally, an estimated 11 million people fell ill with TB (incidence) in 2021.
- Historically, it has been the top infectious disease killer. In 2021, there were an estimated 1.4 million TB deaths and an additional 187 000 deaths among HIV-positive people.
- Three countries accounted for 42% of global cases in 2021: India (26%), the Russian Federation (8.5%) and Pakistan (7.9%).
Image
Figure: Estimated TB incidence in 2021, for countries with at least 100 000 incident cases; Source: Global TB Report, 2022.
Resources
Burden of TB in India
ContentTB is one of the top burdensome infectious diseases in India. It is estimated that, around 1/4th (26%) of the world's TB cases are in India, translating to about 30 Lakhs new TB cases emerging each year (TB incidence). Against this estimated incidence the National TB Elimination program reported around 19 lakh new and relapse cases in the year 2021.
An estimated 5 Lakhs deaths occur due to TB each year in the country, translating to about 1 case of TB death every one-two minutes. Compared to this, there are only about 60 thousand deaths due to HIV and about 77 deaths due to Malaria each year.
TB diagnosis and treatment services although provided free of cost in the public sector, the cost of accessing these services and related loss of wages drive the affected people with poverty (catastrophic costs). TB also has a huge impact on the world's and the country's economy because of loss of workdays (100 million workdays per year).
Assessment
Question Answer 1 Answer 2 Answer 3 Answer 4 Correct answer Correct explanation Page id Part of Pre-test Part of Post-test What is the estimated number of incident TB cases that emerge each year in India? 35 Lakh cases 26 Lakh Cases 26% of the Global Burden 19 Lakh Cases 2 The estimated number of new and relapse (incident) cases in India that emerge each year is about 26 Lakh Yes Yes How many cases of deaths are estimated to be caused by TB in India Approximately One death every 2-3 minutes Approximately 5 Lakh deaths 60 Thousand deaths each year 1 and 2 4 In India it is estimated that there is around one death caused due to TB every one to two minutes, translating to about 5Lakh deaths each year in India Yes Yes Resources:
TB Causative organism
Content
Figure Mycobacterium tuberculosis
TB is caused due to the infection by a bacterium called Mycobacterium tuberculosis.

Figure: Extra-Pulmonary Tuberculosis
It often affects the lungs, and in such cases it is called Pulmonary Tuberculosis. But, it can affect almost any part of the body (except the hair and the nails), in which it is known as Extra-Pulmonary Tuberculosis.
Resources:
Mode of TB Transmission
ContentTuberculosis is transmitted mainly through the air via droplet nuclei generated when a TB patient coughs or sneezes.
It is estimated that every sputum smear-positive patient spreads the infection to 10 – 15 persons annually, if untreated..

Figure: Transmission of TB bacteria through air via droplet
Resources:
- Technical and Operational Guidelines for TB Control in India 2016
- WHO - Fact sheet details on Tuberculosis
Kindly provide your valuable feedback on the page to the link provided HERE
Determinants of TB Disease
ContentDeterminants are any characteristics that affect the health of a patient.
Biological Determinants Behavioral Determinants Socio Economic Determinants Occupational Determinants - People living with HIV(PL HIV)
- History of contact with a case of TB
- People with underlying medical conditions like Diabetes, Kidney disease, Cancer etc.
- Existing lung disease
- Old age
- Use of tobacco and alcohol
- Malnutrition
- Person in contact with TB infected patient
- Person living in areas with poor ventilation & over crowding
- Poverty and Malnutrition
- Homeless
- Mining work
- Quarry work(Silicosis)
- Construction work
- Migrant worker
- Daily wagers
Vulnerable Population for Tuberculosis
ContentTB can affect anyone but it is more prevalent in some communities which are vulnerable to TB disease due to various factors which are mentioned below:
Increased exposure of TB due to where they live or work
- prisoners
- slum dwellers
- miners
- hospital visitors
- healthcare workers
Limited access to Quality TB services
- Migrant workers
- Women in settings with gender disparity,
- Children
- Physically challenged
- Transgender population
- Tribal and population living in hard to reach areas
- Refugees or internally displaced people
- Illegal miners and undocumented migrants
Increased risk because of biological or behavioural factors that compromise immune functions in people who:
- People who live with HIV
- have diabetes or silicosis
- undergo immunosuppressive therapy
- are undernourished
- use tobacco
- suffer from alcohol use disorders.
- inject drugs
TB Infection
Content-
TB Infection (or previously known as Latent TB infection) is a stage in between uninfected and having active TB. In this stage the person has no symptoms and can only be identified using laboratory tests.
-
The vast majority of infected people may never develop TB disease. However, to achieve TB elimination, it is important to treat TB infection in people at risk of developing active TB disease.
-
It is a state of persistent immune response to stimulation by Mycobacterium tuberculosis antigens with no evidence of clinically manifested active TB.
-
There is no single acceptable/reliable test for direct identification of Mycobacterium tuberculosis infection in humans. Tuberculin Skin Test (TST) and Interferon-gamma release assay (IGRA) are commonly used tests for identifying TB infection.
Resources:
-
Progression to TB Disease
ContentAfter exposure to infective droplets containing M.TB, only a small proportion gets infected and further progresses to active TB disease.
- Majority of those that get infected persist in a stage of clinical latency known as TB infection (previously known as Latent TB infection). They do not have TB disease and do not show any symptoms of TB and no evidence of any TB related changes on chest X-ray.
- A small proportion of those with prior infection may progress to active TB disease due to various environmental/ agent/ host factors.

Figure: Flow chart for TB disease progression
Resources:
Kindly provide your valuable feedback on the page to the link provided HERE
TB Infection Vs Active TB Disease
ContentTB Infection Active TB Disease May not have any signs & symptoms Has sign and symptoms such as cough for more than two weeks, fever, weight loss and blood in sputum Has dormant, contained bacteria is the body Has active, multiplying bacteria in the body Doesn't spread TB bacteria to others May spread TB bacteria to others Chest X-ray usually normal Lesion in Chest X- ray (usually) May advance to active TB. It is estimated that the lifetime risk of an individual with TB infection for progression to active TB is 5–10%. Needs treatment for TB disease Resources:
Chemotherapy and its implication in TB control
ContentChemotherapy for TB is the use of an anti-TB drugs to kill, or prevent the replication of, TB mycobacteria in the patient’s body. Effective anti-TB chemotherapy renders the patient non-contagious and cures the patient, thereby interrupting the chain of transmission. Mortality rates of TB range from 50-80% in untreated smear-positive individuals and drop to lower than 5% under chemotherapy.
Most of the bacteria are killed during the first 8 weeks of treatment; however, there are persistent organisms that require longer treatment. TB disease must be treated for at least 6 months and in some cases even longer. The use of multi-drug therapy reduces the incidence of drug-resistant cases and increases the overall effectiveness of treatment.
If treatment is interrupted, any surviving bacteria may cause the patient to later become ill and infectious again, potentially with drug-resistant disease.
How infectious are tuberculosis patients under chemotherapy?
Under effective chemotherapy, there is a substantial decline in infectiousness in two weeks time, and may not be a major source of risk to any contacts. This decline is indicated by the rapid fall in the number of viable organisms in the sputum, and reduced frequency of coughing.
Types of Chemotherapy in TB
- Preventive Chemotherapy: Regimen for healthy but TB infected persons with a high risk of developing TB, in order to prevent them from developing TB.
- Standard Chemotherapy: Two-phased chemotherapy for an average of 6-8 months based on the combination of at least four major drugs (HRZE) given for 2 months during the initial intensive phase of treatment and followed by a combination of at least 2 drugs given for at least 4 months during the continuation phase of treatment.
- Chemotherapy for Drug-resistant TB: Two-phased chemotherapy varying from 9 - 24 months in patients having demonstrated resistance to drugs used in standard chemotherapy. The regimen varies with the drug to which the resistance is present, however, each regimen contains a mix of second-line anti-TB drugs including injectables.
Resources
- Training Modules (1-4) for Programme Managers and Medical Officers, 2020.
- Tuberculosis Infectiousness and Host Susceptibility, The Journal of Infectious Diseases, Vol. 216, suppl_6, 2017.
- Tuberculosis chemotherapy: Current Drug Delivery Approaches, Respiratory Research 7, Article no. 118, 2006.
- Tuberculosis Case-finding and Chemotherapy: Questions and Answers, K. Toman.
Assessment
Question Answer 1 Answer 2 Answer 3 Answer 4 Correct answer Correct explanation Page id Part of Pre-test Part of Post-test Which type of chemotherapy regimen of anti-TB drugs is used for infected persons with a high risk of developing TB who have no signs or symptoms of active disease, in order to prevent them from developing TB? First-line anti-TB drugs Preventive Chemotherapy Standard Chemotherapy Chemotherapy for Drug Resistant-TB 2 Preventive chemotherapy regimen of anti-TB drugs is used for infected persons with a high risk of developing TB who have no signs or symptoms of active disease, in order to prevent them from developing TB. Yes Yes Prevention of TB
ContentAs TB is an airborne infection, TB bacteria are released into the air when someone with infectious TB coughs or sneezes. The risk of infection can be reduced by taking simple precautions:
Figure: Measures for control and prevention of tuberculosis
TB Preventive Treatment(TPT) also has a very important role in prevention of TB. Presently, household contacts of sputum-positive TB patients are given TPT upon confirmation of TB infection and ruling our active Tuberculosis.
Resources:
-
Ph-Ch2: NTEP
FullscreenEvolution of TB Elimination Programme in India
ContentThe National Tuberculosis Control Program (NTP) of India was launched in 1962. It relied on BCG, X-ray based diagnosis and Streptomycin and INH based treatment centralized at district level.
Based on a review of the NTP, and WHO recommendations of the DOTS Strategy, Government of India then revised the NTP and launched new program with the title Revised National Tuberculosis Control Program (RNTCP) in 1997. It used Sputum microscopy at DMC(Designated Microscopy Centres) for diagnosis, and multi-drug Short Course Anti-TB Therapy, decentralized to the TU (TB Unit) level.
In recognition of the rising drug resistance problem the DOTS Plus/ PMDT (Programmatic Management of Drug Resistant TB) was launched in 2006 and scaled up to the entire country by 2012.
Further to strengthen the monitoring and supervision system - a case based notification system - Nikshay was introduced in 2012. The same year Tuberculosis was added as a notifiable disease at the point of diagnosis by all health care providers.
Other key milestones from 2012 to 2020 were the availability of the Standards of TB Care in India (STCI) in 2014, introduction of the Daily weight band wise Fixed Dose combination (FDC) in 2016 and new drugs like Bedaquilline and Delaminid were started in 2017 and 2018 respectively.
To emphasise the commitment of the Government of India and to accelerate the efforts towards TB elimination, RNTCP was renamed as "National Tuberculosis Elimination Programme (NTEP)" in 2020.

Figure: Key milestones under NTEP
Resources:
India's commitment to End TB
ContentThe Government of India has committed to achieving the Sustainable Development Goals(SDG) targets related to ending TB by 2025 (5 years ahead of the global target). This would mean that in 2025, the 2030 target of achieving 80% reduction in incidence, 90% reduction in deaths due to TB compared to that of 2015, is to be achieved.
Parameter 2015 Value SDG 2030 Target Commitment for 2025 Estimated annual Incidence 217 cases/Lakh 80% reduction 44 cases/lakh Estimated annual Mortality 4.5 lakh 90% reduction 45,000 Table: India's commitment to End TB by 2025.
Resources:
NTEP Objectives- in relation to NSP 2017-2025
ContentNSP 2012 - 2017 had the aim of achieving universal access to quality diagnosis and treatment. The NSP 2017-2025 which builds on the success and learnings of the last NSP, and articulates the bold and innovative steps required to move towards TB elimination. In 2020, RNTCP was renamed to "National Tuberculosis Elimination Programme" with the following objectives:
Figure: Objectives of NTEP
Resources:
Organizational Structure of NTEP
ContentNational Tuberculosis Elimination Programme (NTEP) is a centrally sponsored programme being implemented under the aegis of National Health Mission.
National Level: Managed by Central TB Division (CTD), the technical arm of the Ministry of Health and Family Welfare (MOHFW)
State Level: State TB Cell coordinates the overall TB elimination programme in state under the guidance of State Health Society. The training ,supervision, monitoring and evaluation NTEP at state level are looked after by STDC (State TB Training and Demonstration Centre).
District TB Centre (DTC) is the nodal point for all TB elimination activities in the district under the guidance of the District Health Society.
Tuberculosis Unit (TU) Level: NTEP activities at block/sub-district level are implemented through TU which comprises Designated Medical Officer (MO) supported by two full-time NTEP staff - STS (Senior Treatment Supervisor) & STLS (Senior TB Lab Supervisor).
PHI (Peripheral Health Institute): PHI is a health facility manned by a Medical Officer (MO). Some of the PHIs are also the Tuberculosis Diagnostic Centres, which are the most peripheral level laboratories in the NTEP structure. All the Private Health Facilities like Private Practitioners / Private Hospitals / Clinics / Nursing Homes are also PHI.

Figure: Organisational structure of NTEP
Resources:
The State TB Cell
ContentThe State TB Cell or STC is the state-level implementing structure of the National TB Elimination Program (NTEP). It is the leading institution for management of NTEP activities at the state level.
The STC is a State Government entity that acts as the bridge between the Central and State Governments for implementing the NTEP. It works under the guidance of the Central TB Division (CTD), and it oversees the program implementation at the districts.
- The State TB Cell is supported by the State TB Training and Demonstration Centre (STDC) for its technical functionalities. STDC mainly supports training, supervision and monitoring.
- The nodal laboratory for the State is the Intermediate Reference Laboratory (IRL). This supports quality assurance of the smear microscopy network and laboratory services in the state.
- The STC has a fully operational State Drug Store (SDS) which is responsible for the effective management and uninterrupted supply of good-quality of medicines and other logistics.
Human Resources at the State TB Cell are:
- State Tuberculosis Officer (STO). A dedicated official from the state health system, at the rank of a Joint Director is designated as the STO and heads the implementation of the NTEP at state level.
- Medical Officer STC (MO-STC): A medical officer from the state health system assists the STO in overseeing various activities.
- State DRTB Coordinator: Assist the STO in DRTB activities monitoring across the districts
- TB - HIV Coordinator: Assist the STO in overseeing TB comorbidities across the district.
- State PPM Coordinator: Looks at the private sector engagement
- State IEC Officer/ACSM Officer: Oversees the implementation of advocacy, communication and social mobilisation activities across different districts.
- STC - Epidemiologist: Assist the STO and STDC Directors by analyzing state-level data and preparing review materials
- Other support staff at the STC include
- Accounts Officer
- Technical Officer-PSM
- Secretarial Assistant
- Data entry operators/Nikshay operator
Resources
- Training Modules (1-4) for Programme Managers and Medical Officers, 2020.
- Training Modules (5-9) for Programme Managers and Medical Officers, 2020.
Assessment
Question Answer 1 Answer 2 Answer 3 Answer 4 Correct answer Correct explanation Page id Part of Pre-test Part of Post-test Which of the following statements are true about the State TB Cell (STC)? STC is a state government entity. It is the leading institution for the management of NTEP activities at the state level. It is supported by the STDC. All of the above 4 The STC is a state government entity that is the leading institution for the management of NTEP activities at the state level and is supported by the STDC. Yes Yes State TB Training and Demonstration Centre [STDC]
ContentThe State Tuberculosis (TB) Cell (STC) is supported by the State TB Training and Demonstration Centre (STDC) in many states through its three units – Training Unit, Supervision and Monitoring Unit and an Intermediate Reference Laboratory (IRL). This relationship is shown in the figure below.
- Training Unit: It is involved in estimating the training load, organizing state level training (Induction and Refresher) and evaluating the performance of those who undergo training.
- Supervision and Monitoring Unit: It consists of a team which is dedicated to the supervision of TB elimination activities through supervisory visits, periodic desk review of Nikshay and Nikshay Aushadhi data, and plans state internal evaluations apart from assisting in other supervision and monitoring activities of National Reference Laboratories, Central TB Division and other national/international monitoring missions.
- Intermediate Reference Laboratory: This supports an effective quality assurance system of the sputum smear microscopy network and laboratory services for the programmatic management of drug-resistant TB (molecular drug resistance and culture and drug susceptibility testing) in the state.
The STDC is also involved in operational research.
Human Resources in the STDC
- The STDC functions under the leadership of STDC Director.
Training and Supervision & Monitoring Units:
- 1 Epidemiologist
- 1/more Medical Officer
- 1 Nikshay Operator
- 1 Secretarial Assistant
Intermediate Reference Laboratory (IRL):
- 1 Microbiologist
- 1 Microbiologist- External Quality Assistance (EQA)
- 1 Senior Laboratory Technician- EQA
Resources
- Training Modules (1-4) for Programme Managers and Medical Officers, 2020.
- Training Modules (5-9) for Programme Managers and Medical Officers, 2020.
Kindly provide your valuable feedback on the page to the link provided HERE
District TB Centre [DTC]
ContentThe key level for the management of public health services is the district level. The District Tuberculosis Centre (DTC) is the nodal point for tuberculosis (TB) control activities in the district.
Functions of the DTC
The primary role of the DTC is a managerial one. The DTC is the central program management unit of the district responsible for all activities related to National TB Elimination Programme (NTEP) implementation such as:
- Advocacy
- Active case finding
- Diagnosis, treatment (both for drug-susceptible and drug-resistant TB cases) and follow up
- Managing comorbidities
- Service delivery
- Maintaining diagnostic and treatment infrastructure
- Setting up Drug-resistant TB (DR-TB) centres
- Ensuring community engagement and TB forums
- Multi-sectorial involvement for drug management, and supervision and monitoring
- Financial management
- Drugs, logistics and supply chain management.
Components of the DTC
- District Drug Store (DDS)
- Nucleic Acid Amplification Test machine (Cartridge Based NAAT or TrueNAT)
- Designated Microscopic Center (DMC)
- Treatment Support Center
- Drug Resistant TB (DR-TB) Center
- X-Ray Unit
With expansion of TB services and ongoing collaboration with various national programs, the structure of DTC is highly integrated as part of general health system and some components may cater to non-TB patients as well e.g., the DMC may be a part of general laboratory, and X-ray unit can be functional for all departments and not just chest/TB section.
Human Resources Deployed at the DTC
The Chief District Health Officer (CDHO) / Chief District Medical Officer (CDMO) / Civil Surgeon or an equivalent functionary in the district is responsible for all medical and public health activities including control of TB.
A full-time District TB Officer (DTO), trained at the national level and based at the DTC, is responsible for planning, training, supervising and monitoring the programme in the district. The DTO is assisted by other technical and secretarial staff:
- Medical Officer- District TB Center
- District DR-TB-HIV Coordinator
- District Public Private Mix Coordinator
- District Program Coordinator
- District Drug Store Pharmacist
- District Data Entry Operator-Nikshay
- District Accountant
- Senior TB laboratory Supervisor
- Senior Treatment Supervisor
- Laboratory Technicians for DMC and NAAT site
- Counsellor for District DR-TB center
- TB Health Visitors
While the National TB Elimination Program (NTEP) approves the above positions through National Health Mission NTEP Project Implementation Plan, the district always has the flexibility for additional resource deployment based on the need and existing epidemic. The DTO and his/her team are supported by various other program officers/staff and non-governmental organizations working in the field for Tuberculosis and Health.
Resources
- NTEP Training Modules 1-4 for Programme Managers & Medical Officers, 2020.
- NTEP Training Modules 5-9 for Programme Managers & Medical Officers, 2020.
Kindly provide your valuable feedback on the page to the link provided HERE
Tuberculosis Unit [TU]
ContentTuberculosis (TB) unit (TU) is the sub-district level supervisory unit of National TB Elimination Program with the following organogram:
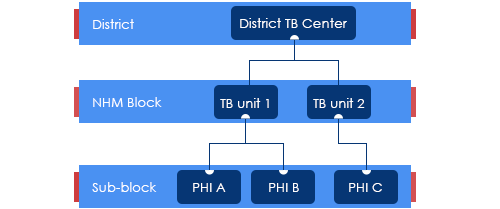
Figure: Organogram of a TB Unit
(PHI: Peripheral Health Institution)
TUs are based mainly on National Health Mission (NHM) health blocks with the aim of aligning with the NHM Block Programme Management Unit (BPMU) for optimum resource utilization and appropriate monitoring.
The TUs have been created based on a population of 1 per 2,00,000 (range 1.5 – 2.5 lakh) for rural and urban populations and 1 per 1,00,000 (0.75 – 1.25 lakh) population in hilly/tribal/difficult areas.
The TU consists of a designated Medical Officer-Tuberculosis Control (MO-TC), as well as one full-time supervisory staff - Senior Treatment Supervisor (STS). However, one Senior TB Laboratory Supervisor (STLS) will be there in every 5 lakh population (one per 2.5 lakh population for tribal/hilly/difficult areas), mostly covering 2-3 TUs.
TB Unit manages the provision of TB services (Diagnosis, Treatment, Prevention, etc.) and programme management in the assigned geographical area.
Resources
Peripheral Health Institutions [PHI] and Health Facilities
ContentUnder the National Tuberculosis Elimination Programme (NTEP), a Peripheral Health Institute (PHI) is a health facility that is manned by at least a Medical Officer (MO), where diagnosis and management of Tuberculosis (TB) are done.
At this level, there are dispensaries, Primary Health Centres (PHCs), Community Health Centres (CHCs), referral hospitals, major hospitals, speciality clinics or hospitals (including other health facilities), TB hospitals, Anti-retroviral Treatment (ART) centres and medical colleges within the respective district.
All health facilities in the private and Non-government Organisation (NGO) sectors participating in NTEP are also considered PHIs. Some of these PHIs also function as Designated Microscopy Centres (DMCs).
Role of PHIs in Program Management for TB Elimination
- PHIs undertake tuberculosis case-finding and treatment activities as a part of the general health services.
- In situations where more than one MO is posted in any of the PHC, one of them may be identified and entrusted with the responsibilities of the NTEP.
- Additionally, NTEP provides 1 TB Health Visitor (TBHV) per one lakh urban population to support the urban TB control activities in urban settings/ medical colleges.
Resources
Drugs Stores in NTEP
ContentImage
DRUG STORE
Central TB Division, MoHFW, has
Under NTEP, there is a large network of drug stores across the country to ensure a regular and uninterrupted supply of drugs and consumables. The Drugs and consumables are procured at the Central level and supplied at Central warehouses (GMSDs & CMSS); further drugs and consumables are supplied to the State Drug Stores and further dissemination to district and sub-district levels following the stocking norms to ensure uninterrupted supply of drugs and consumables to the patient.
To provide overall policy guidance and coordination, the Procurement and Supply Chain Management (PSM) Unit has been established at Central TB Division (CTD), MoHFW, for procurement and Supply Chain Management of all types of anti-TB drugs, diagnostics and consumables.
Standards of TB Care in India
ContentThe Standards for TB Care in India (STCI), which is a locally customized version of the International Standards of Tuberculosis Care, mentions 26 standards that every citizen of India should receive irrespective of the sector of treatment.
STCI were developed based on a series of discussions involving various stakeholders including clinicians, public health specialists, community workers and patient advocates.
STCI represent what is expected for quality TB care from the Indian healthcare system including both public and private systems.
It was first published in 2014 and outlines standards across the four themes of TB diagnosis, TB treatment, public health action and social inclusion.
Following are the list of the 26 Standards:

Table 1: Categorisation of the Standards for TB Care in India, Source: Standards for TB Care in India, World Health Organisation, pp. 13-23
Resources
Assessment
Question Answer 1 Answer 2 Answer 3 Answer 4 Correct answer Correct explanation Page id Part of Pre-test Part of Post-test How many standards in TB care are described in the Standards of TB Care in India (STCI) 2014? 4 15 26 32 3 There are 26 standards for TB care under four major categories: diagnosis, treatment, public health actions and social inclusion. Yes Yes Stages in TB Patient's Lifecycle
ContentThose who are suspected of having TB disease are first screened for symptoms like cough and fever for more than 2 weeks, blood stained sputum and weight-loss. If found positive on screening, then TB patients are referred for testing to the nearest health facility. If diagnosed with TB, then they are subsequently initiated on treatment. The TB patients initiated on treatment are regularly monitored with the help of field staff or digital interventions like 99DOTS and MERM (Medication Event Reminder Monitor) technology. NTEP staff also ensures that the TB patients are regularly followed up on monthly basis till their treatment completion.

Figure: Patient Flow
Kindly provide your valuable feedback on the page to the link provided HERE
-
Ph-Ch3: Diagnostic Technologies
FullscreenTesting for TB diagnosis
ContentNational Tuberculosis Elimination Programme (NTEP) strives for all presumptive TB patients to be microbiologically confirmed. Under NTEP, the acceptable methods for microbiological diagnosis of TB are:
Sputum Smear Microscopy (for Acid Fast Bacilli - AFB): Sputum Smear microscopy is the primary tool which is reliable, inexpensive, easily accessible and rapid method of diagnosing PTB, where in the bacilli are demonstrated in the sputum. Two types:
-
Ziehl-Neelsen Staining
-
Fluorescence staining
Rapid diagnostic molecular test: Rapid molecular tests that use techniques like NAAT are very specific. They amplify the genomic material in the patient sample and hence enhances detection
-
Nucleic Acid Amplification Test (NAAT) e.g., GeneXpert, TrueNat
Figure: Genxpert Machine for CBNAAT
Figure: Truenat Machine
- Line Probe Assay
Culture and DST: A culture test involves studying bacteria by growing the bacteria on different substances. This is to find out if particular bacteria are present. In the case of the TB culture test, the test is to see if the TB bacteria Mycobacterium tuberculosis, are present.
Two types:
-
Solid (Lowenstein Jensen) media
-
Liquid media (Middlebrook) e.g., Bactec MGIT etc.
-
Microscopy
ContentMicroscopy is a TB diagnostic technology that utilizes the acid-fastness property of Mycobacterium tuberculosis to visualize it under a microscope. Results of sputum smear microscopy can either be smear-negative, or smear-positive (with various grades).
Advantages:
- It is currently the most accessible and cheapest TB diagnostic test available under National TB Elimination Programme (NTEP) in India.
- It has the shortest turnaround time for diagnosis.
- It has high specificity.
Limitations:
- Low sensitivity. It becomes positive only when more than 5000 bacilli/ml of sample are present. Hence, cases would be missed in early disease, or when an inappropriate biological specimen is provided, where bacterial load in sputum is less.
- It is unable to differentiate between M. tuberculosis and Non-tuberculous Mycobacteria (NTM). This is predominantly an issue in geographies with lower burden.
There are two types of microscopies used in NTEP: Ziehl-Neelsen (ZN) Microscopy and Fluorescence Microscopy (FM). These vary in the type of stain and microscope used. FM is newer of the two types and is currently recommended for use over ZN.
Resources
Cartridge Based Nucleic Acid Amplification Test [CBNAAT]
ContentCartridge Based Nucleic Acid Amplification Test (CBNAAT) is a rapid molecular diagnostic test. It is used for diagnosis of Tuberculosis (TB) and Rif-resistant Tuberculosis (RR-TB) in NTEP. Results are obtained from unprocessed sputum samples in about 2hours which helps in early detection and treatment of TB patients.
India has vast number of CBNAAT laboratories which are utilized for TB/RR-TB detection and Universal Drug Susceptibility Testing (UDST) under the National TB Elimination Program (NTEP).

Figure: CBNAAT Cartridge and Machine in Use (Image courtesy: USAID supported Challenge TB Project)
The CB-NAAT system detects DNA sequences specific for Mycobacterium tuberculosis complex and rifampicin resistance by Polymerase Chain Reaction (PCR). It concentrates Mycobacterium tuberculosis bacilli from sputum samples, isolates genomic material from the captured bacteria by sonication and subsequently amplifies the genomic DNA by PCR. The process identifies clinically relevant rifampicin resistance-inducing mutations in the RNA polymerase beta (rpoB) gene in the Mycobacterium tuberculosis genome in a real-time format using fluorescent probes called molecular beacons.
Video fileVideo: Cartridge-Based Nucleic Acid Amplification Test [CBNAAT] - GeneXpert Technology
Resources
- Training Module (1-4) for Program Managers and Medical Officers, NTEP, MoHFW, 2020.
- India TB Report 2021, National TB Elimination Program (NTEP), MoHFW, 2021.
Assessment Questions
Question
Answer 1
Answer 2
Answer3
Answer 4
Correct Answer
Correct explanation
Part of pre-test
Part of post-test
Under NTEP, CBNAAT is offered upfront for which of these categories?
PLHIV
Paediatric presumptive TB
Presumptive DR-TB
All of the above
4
Under NTEP, CBNAAT is recommended upfront for People living with HIV, Paediatric Presumptive TB patients, Presumptive DR-TB patients and patients notified from the Private sector.
Yes
Yes
CBNAAT requires the processing of sputum samples before testing
True
False
2
Results are obtained from unprocessed sputum samples in about 2hours from a CBNAAT machine
Yes
Yes
Intended Use of TrueNAT MTB and TrueNAT MTB Rif Dx Assay
ContentTrueNAT MTB and TrueNAT MTB Rif Dx Assay are intended to be used for detection of Mycobacterium tuberculosis bacteria (for TB diagnosis) and to detect resistance to rifampicin (Rif), respectively.
Positioning of TrueNAT MTB and TrueNAT MTB Rif Dx Assay
TrueNAT MTB and TrueNAT MTB Rif Dx Assay are suitable as point-of-care tests for use in peripheral laboratories and microscopy centres due to the following operational features:
- Biosafety requirements similar to smear microscopy
- Sample pre-treatment and preparation kits stable at 2-40°C, while TrueNAT chips stable between 2-30°C
- Test reagents with shelf life of 2 years
- Operates up to 8 hours on fully-charged batteries
- Temperature controlled operation, like air-conditioning, not required (operates up to ≤ 40°C)
- Operates under a wide relative humidity range (10-80%) & dusty conditions
- Portable and comes with a carrying case.
Target population
National TB Elimination Programme (NTEP) has defined the use of Nucleic Acid Amplification Test (NAAT) like Cartridge Based Nucleic Acid Amplification Test (CBNAAT) and TrueNAT as upfront tests for diagnosis of TB in presumptive paediatric TB, presumptive extra-pulmonary cases, People Living with HIV (PLHIV), smear negative X-ray suggestive of TB, contacts of Drug Resistant TB (DR-TB), treatment non-responders, TB affected patients retrieved after loss to follow-up and other vulnerable groups.
Resources
- Guidelines for Programmatic Management of Drug resistant Tuberculosis (PMDT) in India, 2021.
- Practical Guide to Implementation of TrueNAT Tests for the Detection of TB and Rifampicin Resistance.
Kindly provide your valuable feedback on the page to the link provided HERE
Line Probe Assay [LPA]
ContentLine Probe Assay (LPA) is a rapid molecular test available at centralised laboratories.
The assay is based on Polymerase Chain Reaction (PCR) that can simultaneously detect Mycobacterium tuberculosis complex as well as drug sensitivity to anti-TB drugs.

Figure 1: The GenoType MTBDRplus Molecular LPA Procedure; Source: Molecular Detection of Drug-resistant Tuberculosis by Line Probe Assay.
Advantages of LPA
- Rapid molecular test. (Turnaround time: 3-5 days)
- Highly sensitive and specific.
- Performed directly from sputum smear-positive specimens and on isolates of M. tuberculosis complex grown from smear-negative and smear-positive specimens.
- Detects multiple gene mutations in anti-TB drugs.
- First-line LPA detects mutations to rifampicin and isoniazid
- Second-line LPA detects mutations to fluoroquinolones and aminoglycosides.
- Suitable for low and high-throughput labs.
Disadvantages of LPA
- Cannot be used as a point-of-care test.
- Requires appropriate laboratory infrastructure, equipment and biosafety precautions.
- Different rooms (DNA extraction, pre-amplification, amplification, post-amplification/ hybridization) are required to perform different steps (Figure 2).
- Requires trained manpower to perform tests and interpret test results.
- Stringent internal quality control is required to prevent contamination.

Figure 2: Amplification (A) and Post-amplification Laboratory (B) for LPA; Source: Molecular Detection of Drug-resistant Tuberculosis by Line Probe Assay.
Resources
- Guidelines for PMDT in India, 2021.
- Molecular Detection of Drug-resistant Tuberculosis by Line Probe Assay.
Assessment
Question Answer 1 Answer 2 Answer 3 Answer 4 Correct answer Correct explanation Page id Part of Pre-test Part of Post-test LPA can be used as a point-of-care test. True False 2 LPA cannot be used as a point-of-care test. Yes Yes Solid and Liquid Culture in TB
ContentCulturing TB Bacilli is well known and historic method for detection/ confirmation of Tuberculosis. It is a highly sensitive and specific phenotypic test; it can detect even a few viable bacilli in the sample (Upto 10 Colony Forming Units- CFUs). TB bacilli multiply in the culture and form colonies of TB bacilli which can are easily be identified.
Based on the growth media used Culture is divided in to two types, Solid and Liquid Culture methods. Types Culture:
- Solid Culture on Lowenstein Jensen media : Historic gold standard culture test. Results take usually upto 2 months (60 days).
- Modern Liquid culture systems: (e.g. BACTEC MGIT 960, BacT Alert or Versatrek etc.) Results take usually up to 42 days.
Uses
- Solid culture is the gold standard diagnostic test for TB. But it is not used for the purpose of TB diagnosis due to the long turn around time of 2 months. It is largely used for research purposes where it is used as the baseline test on which the sensitivity and specificity of other tests are calculated.
- Liquid Culture is being used for follow-up monitoring of patients on drug resistant TB treatment to detect treatment failure. Liquid culture is also used for long term follow up patients who have successfully completed treatment to detect recurrence.
- Liquid culture is used as a previous step to grow bacilli and obtain isolates prior to Drug Susceptibility Testing.
- Liquid cultures are also used in TB prevalence surveys for its high sensitivity and specificity
Resources
Kindly provide your valuable feedback on the page to the link provided HERE
Culture Drug Susceptibility Testing [CDST]
ContentCulture Drug Susceptibility Testing (CDST) is a growth-based phenotypic method used to check the susceptibility of Mycobacterium tuberculosis strains to various first and second line anti-TB drugs. Mycobacterial resistance to a particular drug is identified if there is growth observed in culture in presence of that drug.
In NTEP CDST is the standard method to detect resistance in samples of patients who have tested positive on followup. While CDST is possible on both Solid and Liquid culture, currently, the NTEP utilizes only liquid culture as a method for DST, due to faster Turn around times.
CDST testing services are available under NTEP in designated, specialized laboratories called CDST Labs both in public and private sector. Currently there are 80 such laboratories (60 certified for First Line and 49 for Second line drugs). Such designated laboratories are subject to regular external quality assessment, often by the National Reference Laboratory at that region.
Quality assured DST to R, H, Z, Mfx, Lfx, Lzd, Am, Km and Cm are available across the country.
Resources
-
Ph-Ch4: Approaches to TB Case Finding
FullscreenScreening For Tuberculosis Disease
ContentScreening for active tuberculosis (TB) a process to filter out people who are less likely to have TB, from a group. Screened positive people are likely to have TB and are confirmed subsequently using a TB diagnostic test. This will allow finite diagnostic testing resources to be used on the remaining.
Screening in TB may be performed using simple field tools (4 Symptom complex) and tests such as Chest X-ray, or a combination of both. Combination of both is the most effective, but is often not applied due to the practical difficulties in making a chest X-ray conveniently available.
Screening is an integral part of any general case finding effort. It is also applied systematically in specific situations.
- At health care facilities (intensified case finding): Here those visiting are screened using the 4 symptom complex, often at the point of entry to the facility. Those screened positive may be fast-tracked to TB Diagnostic testing.
- In vulnerable populations in active case finding efforts: Here the entire population identified for active case finding are screened using the pre-decided protocols by going door to door.
Resources
Active Case Finding
ContentSystematic screening of all individuals of a defined population is known as active case finding. It is applied outside of health facilities at the community level by the health system.
Objective of ACF is to:
- identify cases early, initiate prompt treatment, reduce risk of poor treatment outcomes and reduce risk of further transmission of TB
- to provide access to diagnosis services to populations that would have been otherwise unreached
It is effort intensive and is recommended only in population groups where there is estimated high case load. In NTEP, ACF is recommended only to be performed in Key / vulnerable population.
ACF can also be clubbed with suitable ACSM campaigns to create awareness about the signs and symptoms and about TB in the target population/ community. It can also be combined with other health activities/ campaigns (such as Pulse Polio/ Leprosy screening/ population based screening for NCDs) for increased efficiency.
Resources
- Training Modules for Programme Managers and Medical Officers.
- Active TB Case Finding, Guidance Document.
- WHO recommendations for Systematic Screening for Active Tuberculosis
Assessment
Question Answer 1 Answer 2 Answer 3 Answer 4 Correct answer Correct explanation Page id Part of Pre-test Part of Post-test Which of the following is not a primary objective of ACF? Increase TB notification Early identification of cases. Reduce the risk of transmission of TB. Reduce the risk of poor treatment outcomes. 1 Notification is not a primary objective of ACF. Yes Yes
Fullscreen