Principle of MGIT DST Reading
ContentPrinciple of MGIT DST Reading:
Two Mycobacterial Growth Indicator Tubes (MGITs) are inoculated with the test culture. Then, a known concentration of a test drug is added to one of the MGIT tubes (drug-containing tube), and growth is compared with the MGIT tube without the drug (growth control).
If the test drug is active against the isolated mycobacteria, it will inhibit the growth, and thus there will be suppression of fluorescence, while the growth control will grow uninhibited and will have increasing fluorescence.
The BACTEC MGIT 960 instrument continually monitors all tubes for increased fluorescence. Analysis of fluorescence in the drug-containing tubes compared to the fluorescence in the Growth Control tube is used to determine susceptibility results.
When the growth unit (GU) of the growth control reaches 400 within 4-13 days (SIRE) or 4-21 days (PZA), the GU values of the drug-containing vials are evaluated.
• S = Susceptible = the GU of the drug tube is less than 100
• R = Resistant = the GU of the drug tube is 100 or more
Resources
Assessment
Answer 1
Answer 2
Answer 3 Answer 4 Correct answer
Correct explanation
Page id
Part of Pre-test
Part of Post-test
Drug-containing vials are evaluated when the growth unit (GU) of the growth control reaches 400.
True
False
1
When the growth unit (GU) of the growth control reaches 400 within 4-13 days (SIRE) or 4-21 days (PZA), the GU values of the drug-containing vials are evaluated.
YES
YES
Procedure for Reading MGIT DST Results
ContentProcedure for Reading MGIT DST Results
Once the Antimicrobial Susceptibility Testing (AST) sets are loaded/entered, the BACTEC MGIT 960 instrument continually monitors all tubes for increased fluorescence. When the growth unit (GU) of the Growth Control (GC) tube reaches ≥400 within the timed protocol, the instrument marks the DST as complete and interprets the results. GU values of the drug-containing vials are evaluated, and final results are reported as Susceptible or Resistant as follows:
-
S = Susceptible = the GU of the drug tube is less than 100
-
R = Resistant = the GU of the drug tube is 100 or more
The steps involved in reading the MGIT DST results are listed below:
-
Completed AST sets (indicated by a red “+” on the drawer) are removed from the respective drawers, and an “Unloaded AST Set” report is printed.
-
All the unloaded AST sets are matched with the printed report and any discrepancies observed are resolved.
-
Before reporting, all ‘resistant’ tubes are observed visually for evidence of contamination. ZN stain is performed on any suspicious tube and subculture to a Blood Agar or Brain Heart Infusion agar Plate. Additionally, when drug resistance is observed and the patient’s isolate was not previously resistant to the drug, tube(s) are tested with ZN and Blood Agar or Brain Heart Infusion Agar Plate to ensure that growth is not due to contaminants or MOTT.
-
DST reports are recorded as “Susceptible” or “Resistant” on the internal lab worksheet/book and the laboratory register.
Resources
Assessment
Question
Answer 1
Answer 2
Answer 3
Answer 4
Correct answer
Correct explanation
Page id
Part of Pre-test
Part of Post-test
The MGIT instrument marks the DST as complete and interprets the results when the GU in the Growth control tube reaches ________.
≥100
≥400
≥200
≥1000
2
When the growth unit (GU) of the Growth Control (GC) tube reaches ≥400 within the timed protocol, the instrument marks the DST as complete and interprets the results.
YES
YES
-
Unloaded MGIT DST Report
ContentUnloaded MGIT DST Report
A completed DST in the Bactec MGIT-960 instrument is indicated by a red “+” on the drawer. The following steps are involved in removing the completed DSTs from the instrument and printing the “Unload AST Set” report:
Step 1: Open the desired drawer. Press the “Remove Completed AST Sets”
soft key. Step 2: The first completed AST set stations illuminate with FLASHING GREEN indicators.
Step 3: Remove the carrier, starting with the completed set closest to the front of the drawer, and scan its barcode label. The LEDs at this station extinguish.
Step 4: Repeat steps 2 – 3 to remove additional AST sets.
Step 5: Place completed AST sets in the AST tube rack.
Step 6: Close the drawer and press the “printer” soft key to access the report selection.
Step 7: Press the “Unloaded AST set report” soft key to print the report.
“Unloaded AST set report” soft key to print the report. Note: Ensure that a working printer is connected to the instrument and check that the paper is present before printing the report.
Resource
Mycobacteriology Laboratory Manual, First Edition, April 2014, Global Laboratory Initiative
Assessment
Question
Answer 1
Answer 2
Answer 3 Answer 4 Correct answer
Correct explanation
Page id
Part of Pre-test
Part of Post-test
A completed DST in the Bactec MGIT-960 instrument is indicated by a red “+” on the drawer.
True
False
1
A completed DST in the Bactec MGIT-960 instrument is indicated by a red “+” on the drawer.
YES
YES
Validation of Resistant/Unexpected MGIT DST Results
ContentValidation of Resistant/Unexpected MGIT DST Results
1. Validation of Resistant MGIT DST Results
Observe all ‘resistant’ tubes visually for evidence of contamination when first removed from the instrument. Then, perform a ZN stain on any suspicious tube and subculture to a Blood agar plate (BAP) or Brain Heart infusion (BHI) agar plate to rule out the contamination.
In addition, when drug resistance is observed, and the patient’s isolate has not been tested before, or if the isolate was not previously resistant to the drug, test the tube(s) with ZN and BAP/BHI to ensure that growth is not due to contaminants or Mycobacteria Other Than Tuberculosis (MOTT).
2. Validation of Unexpected MGIT DST Results
If DST results for any drug are inconsistent with previous results for the same patient, review the results and QC and repeat the test. If the repeat result is discrepant with the first result, repeat the test a third time and record the third test result as the tiebreaker.
Resources
Assessment
Question
Answer 1
Answer 2
Answer 3 Answer 4 Correct answer
Correct explanation
Page id
Part of Pre-test
Part of Post-test
Validation of the results is required if DST results for any drug are inconsistent with previous results for the same patient.
True
False
1
If DST results for any drug are inconsistent with previous results for the same patient, review the results and QC and repeat the test.
YES
YES
If DST results for any drug are inconsistent with previous results for the same patient, review the results and report. True False 2 If DST results for any drug are inconsistent with previous results for the same patient, review the results and QC and repeat the test. If the repeat result is discrepant with the first result, repeat the test a third time and record the third test result as the tiebreaker. YES YES LC DST Recording and Reporting
ContentLC DST Recording and Reporting
LC DST Recording
Once the LC DST results are validated, they should be recorded in the standard ‘NTEP Laboratory Register for Culture, CBNAAT and Drug Susceptibility Testing (Annexure IV)’. The result of the DST for a particular drug is recorded as either S (Sensitive) or R (Resistant) in the box provided for that drug.
A copy of Annexure IV is given below:
ImageLC DST Reporting
NTEP guidelines mandate the reporting of DST results to the requesting district/physician through Ni-kshay within the stipulated turn-around time. Resources:
Guidelines for programmatic Management of Drug-resistant Tuberculosis in India, 2021
Assessment
Question
Answer 1
Answer 2
Answer 3
Answer 4
Correct answer
Correct explanation
Page id
Part of Pre-test
Part of Post-test
The C&DST laboratory records the results of LC-DST in which of the following Annexures?
Annexure I
Annexure II
Annexure V
Annexure IV
4
Once the LC DST results are validated, they should be recorded in the standard ‘NTEP Laboratory Register for Culture, CBNAAT and Drug Susceptibility Testing (Annexure IV)’.
YES
YES
Tuberculosis Laboratory Register
ContentThe Tuberculosis (TB) Laboratory Register is a paper-based recording register kept in all National TB Elimination Programme (NTEP) laboratories for recording details of diagnostic services offered to TB patients referred from both private and public health facilities.
The register is maintained in the Designated Microscopy Centre (DMC). It is the only register used for recording the details of specimen smear examinations. The Laboratory Technician (LT) is responsible for maintaining and updating the laboratory register.
There are two portions in the TB lab register and the table below shows the details captured in each portion.
Table: Pages and Information Covered in the TB Laboratory Register; Source: NTEP Training Module 2 for Programme Managers & Medical Officers
PORTION
DETAILS CONTAINED
Left-hand Portion
Lab serial number assigned by the LT
Date of the sample collection
Patient details such as name, sex, age, address, and contact number
Information on if the patient is from a key population
Referring health facility details
Reasons for examination
Right-hand Portion
Details on the type of specimen
Visual appearance results along with dates
Comorbidity status (HIV and Diabetes)
Details of drug susceptibility testing
Nikshay ID / Notification
Treatment initiation details
The last two columns are for the LT’s signature and remarks by the LT or supervisor
Important Points to Note
- Duplicate registers should not be maintained.
- LTs should ensure that the correct laboratory serial number is recorded.
- Laboratory serial number is given to the patient and not to the sample. A new number should be assigned to every presumptive TB case whose sputum is to be examined.
- All smear-positive (including scanty) results should be recorded in red ink.
- No over writing or manipulation in already entered data should be done.
The figure below shows the left-hand portion of the TB lab register. Click here to access the full form in the NTEP Training Module 2 for Programme Managers & Medical Officers, p. 219.
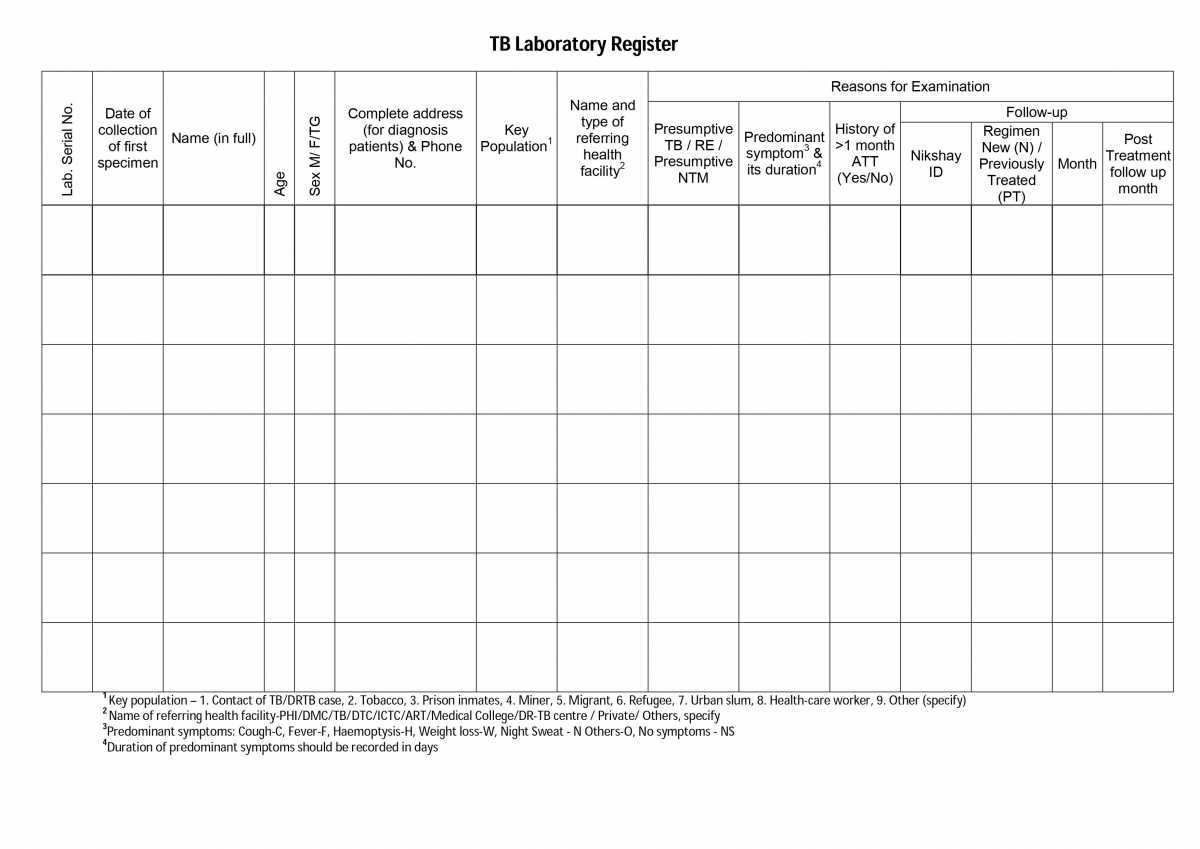
Figure: First Page of the TB Laboratory Register; Source: NTEP Training Modules 1-4 for Programme Managers & Medical Officers, p. 219
Resources
Kindly provide your valuable feedback on the page to the link provided HERE
Drug Susceptibility Testing Logs
ContentMGIT DST testing logs are maintained by the laboratory for QC purposes to record information on the Lot number of MGIT tubes and drugs used along with their expiry dates. A sample DST log is shown here that laboratories can adapt and use as per their requirements:
Image
Resources:
Assessment
Question
Answer 1
Answer 2
Answer 3
Answer 4
Correct answer
Correct explanation
Page id
Part of Pre-test
Part of Post-te
DST logs should contain information on the following:
Lot number of MGIT tube and drugs used Test has to be assigned to a new station NTMs obtained Incubation time for each tube 1 DST logs should contain information on Lot number of MGIT tube and drugs used
Final Result Reporting
ContentFinal Result Reporting
Once the DST results are validated and finalized, these need to be communicated to the requesters. NTEP guidelines mandate the reporting of DST results to the requesting district/physician through Ni-kshay within the stipulated turn-around time. The following steps are involved in reporting the DST results in Ni-kshay:
Step 1: Log in to the Ni-kshay portal and search for the patient whose DST results are being reported by entering the Ni-kshay/episode ID.
Step 2: From the Diagnostic module drop-down, select the ‘add test’ option.
ImageStep 3: Select the appropriate option from the drop-down menu of ‘Reason for testing’ and select ‘DST’ from the ‘Test Type’ drop-down menu.
Step 4: Select the appropriate options in the ‘Facility Details’ section.
ImageStep 5: In the ‘Test details’ sections, after filling in the information about testing and reporting date, select the drug for which the DST is being reported from the drop-down menu of ‘DST to Drug’. Select the Susceptible/Resistant/None (whichever is applicable) option from the drop-down menu of ‘Resistance Status’. Choose the applicable option from the drop-down menu of ‘Final Interpretation’.
ImageStep 6: After adding remarks (if any), hit the ‘submit test’ option to complete the process.
ImageResource
Guidelines for programmatic Management of Drug-Resistant Tuberculosis in India, 2021.
Assessment
Question
Answer 1
Answer 2
Answer 3
Answer 4
Correct answer
Correct explanation
Page id
Part of Pre-test
Part of Post-test
NTEP guidelines mandate the reporting of DST results to the requesting district/physician through which of these?
Request forms
Lab report form
Ni-kshay
Telephone
3
NTEP guidelines mandate the reporting of DST results to the requesting district/physician through Ni-Akshay within the stipulated turn-around time.
YES
YES
Fullscreen