Precautions for Sample Processing and Reagent Preparation in TB Culture Labs
ContentPrecautions for sample processing and reagent preparation in TB culture labs are as follows:
- Precaution is necessary while performing aerosol-generating procedures such as centrifugation, vertexing, mixing, pipetting, pouring and inoculation of media. For example -delay the opening of caps until aerosols have settled, open centrifuge canisters only inside the Bio Safety Cabinets (BSCs), use pipettes that are easy to control.
- Safety precautions to minimize interruption of airflow inside the BSC by keeping arms parallel to the work surface inside the BSC and working in the center to minimize arm movements and ensure not to move hands out of the hood until work is completed. Minimize the equipment inside the BSC so that there is no interference in the airflow pattern.
- Disinfect the BSC and all work surfaces with a tuberculocidal disinfectant before and after every procedure.
- The autoclave should be monitored with a spore test, at least monthly, to ensure that sterility is achieved.
- Avoid practices that can result in spills.
- Train all personnel working in the TB culture lab.
- Working staff must wear Personal Protective Equipment (PPE) like protective gowns, gloves, hair covers and shoe covers etc.
Resources
Kindly provide your valuable feedback on the page to the link provided HERE
BACTEC MGIT 960 Growth Supplement
ContentThe MGIT 960 Growth Supplement (Figure below) is the enrichment added to the MGIT medium prior to inoculation of specimen.
It provides substances essential to the growth of mycobacteria, such as:
- Oleic acid, that is utilized by tubercle bacteria and plays an important role in the metabolism of mycobacteria.
- Albumin, that acts as a protective agent by binding free fatty acids which may be toxic to Mycobacterium species, thereby enhancing their recovery.
- Dextrose, that is an energy source.
- Catalase, that destroys toxic peroxides that may be present in the medium.
- Polyoxyethylene stearate, that enhances the growth of Mycobacterium tuberculosis and assists in providing a uniform inoculum.
Figure: MGIT 960 Growth Supplement
Constituents of MGIT 960 Growth Supplement
15 ml MGIT 960 Growth Supplement contains:
- Bovine Albumin: 50.0 gm
- Dextrose: 20.0 gm
- Catalase: 0.03 gm
- Oleic Acid: 0.1 gm
- Polyoxyethylene state (POES): 1.1 gm
NOTE: It is a sterile product, do not use if turbid or contaminated.
Resources
Kindly provide your valuable feedback on the page to the link provided HERE
Quality Control of MGIT 960 Tubes
ContentMGIT 960 media quality control (QC) is to be done upon receipt of a new shipment or lot number. This includes checking the media visually and by growing control strains of Mycobacterium. Steps include:
- Visually check the tube for:
- Turbidity/contamination
- Expiry date
- Breakage
- Transport condition
- Control organisms prepared in Middlebrook 7H9 Broth should be used for QC testing as per the procedure shown in the figure below. It is important to test all three organisms, as each have different requirements for growth, and each will demonstrate a different time to detection. A good quality control program ensures that the media supports the growth of a variety of organisms, not just one organism.
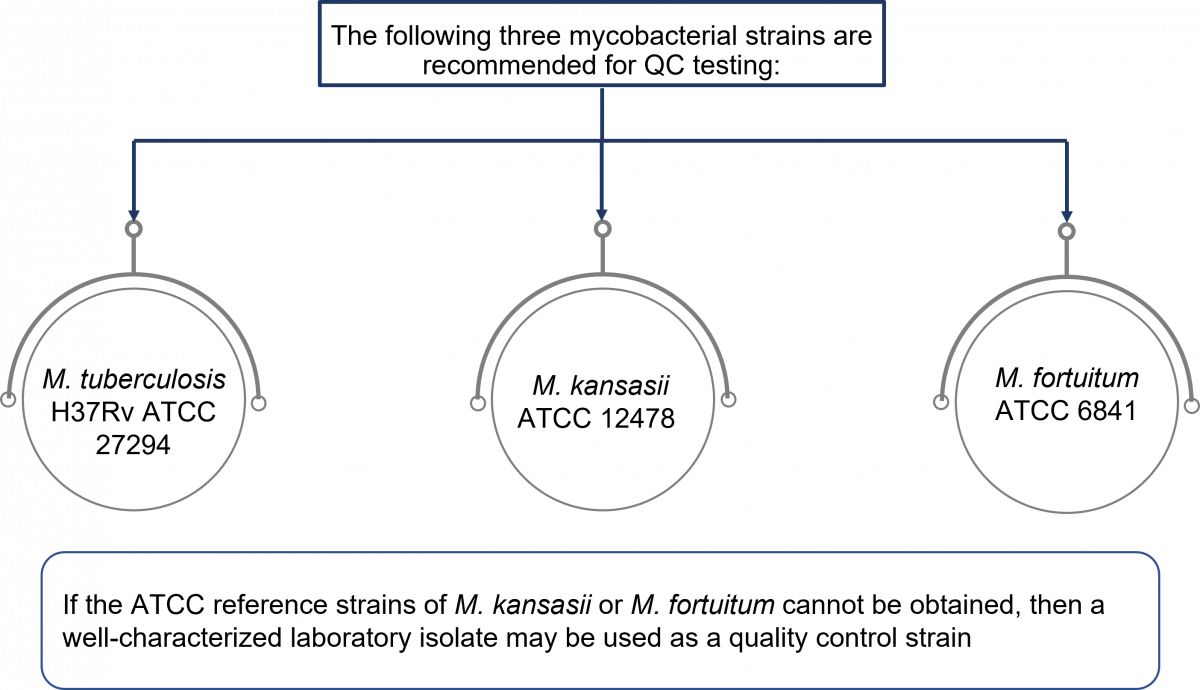
Figure: Control strains used for quality control of MGIT tubes
- Quality control testing should be performed in a biosafety cabinet
-
Subculture the quality control strains on Lowenstein-Jensen (LJ) slants and use pure and fresh growth to prepare a uniform suspension of 0.5 McFarland turbidity
- Dilution of culture suspension using sterile saline or distilled water, accordingly:
- M. tuberculosis - 1:500
- M. fortuitum - 1:5000
- M. kansasii - 1:50000
- Inoculate 0.5 ml of respective dilutions of culture suspension into two labeled MGIT tubes and further incubate the tubes into MGIT 960 system
- The expected results are:
- M. tuberculosis: Positive in 6 to 10 days
- M. kansasii: Positive in 7 to 11 days
- M. fortuitum: Positive in 1 to 3 days
Resources
Kindly provide your valuable feedback on the page to the link provided HERE
- Visually check the tube for:
Fullscreen