Biosafety in TB Laboratories
ContentWhat is Biosafety?
Biosafety is safe handling and containment of infectious microorganisms and hazardous biological materials.
Why Biosafety?
Infections which are obtained through laboratory or laboratory-related activities are known as Laboratory Acquired Infections (LAI). These infections are major occupational health hazards and are a cause of concern for the safety of the staff working in laboratories.
Routes of entry of LAI
There are multiple routes of entry and transfer of toxic material through the body and its transfer to various organs and systems.
The most predominant routes of LAI are:
- Nose - Inhalation of infectious aerosols
- Mouth - Ingestion or exposure through mouth pipetting or touching mouth or eyes with contaminated fingers or contaminated object
- Skin and mucous membrane:
- Spills and splashes onto skin and mucous membranes
- Parenteral inoculations with syringes or other contaminated sharps
- Animal bites and scratches from research laboratories or activities
Biosafety guidelines are prepared to promote:
- Safe microbiological practices
- Safety equipment and facility safeguards for reducing LAIs
- To protect public health and the environment
Biosafety in Tuberculosis TB laboratory (lab)
In a TB lab, the primary risk for infection transmission is the aerosol contaminated with TB bacilli when:
- TB patients walk into the laboratory for specimen collection or other testing and may be a source of droplet infection if they cough during laboratory visits
- Procedures such as centrifugation, vortexing, and vigorous shaking generate significant aerosols that are biohazardous
Key points to consider for M. TB as a biological hazard are given in the table below.
MYCOBACTERIUM TUBERCULOSIS (M. TB) AS BIOLOGICAL HAZARD
RISK FACTORS
Pathogenicity
M.TB exposure may lead to infection
5-10% of infected persons will develop TB disease
Primary route of transmission
Inhalation
Stability
Tubercle bacilli can remain viable for extended periods in the environment
Infectious dose
As little as 10 bacilli can infect humans
Effective Vaccine (for adults)
No vaccine is available for adults
Effective treatment for strain susceptibility to different medicines
Yes
Effective treatment for MUltiple Drug Resistant (MDR), Extensively Drug Resistant (XDR) strains
Yes, but more difficult to treat than susceptible strains
Standards for TB Lab Biosafety
- Standards are prepared based on the recommendations from the WHO expert group in 2012.
- Standards use a procedural approach for the assessment of risk.
- It establishes minimum requirements necessary to ensure biosafety during TB microscopy, culture, drug-susceptibility testing (DST) and molecular testing in different countries and epidemiological settings.
Resources
Kindly provide your valuable feedback on the page to the link provided HERE
Biosafety in TB Laboratory: Risk Assessment Process
ContentRisk assessment is the process that enables the appropriate selection of microbiological practices, safety equipment, and facility safeguards that can prevent laboratory-associated infections (LAIs).
The figure below shows a flowchart that presents the risk assessment process for a biological hazard.
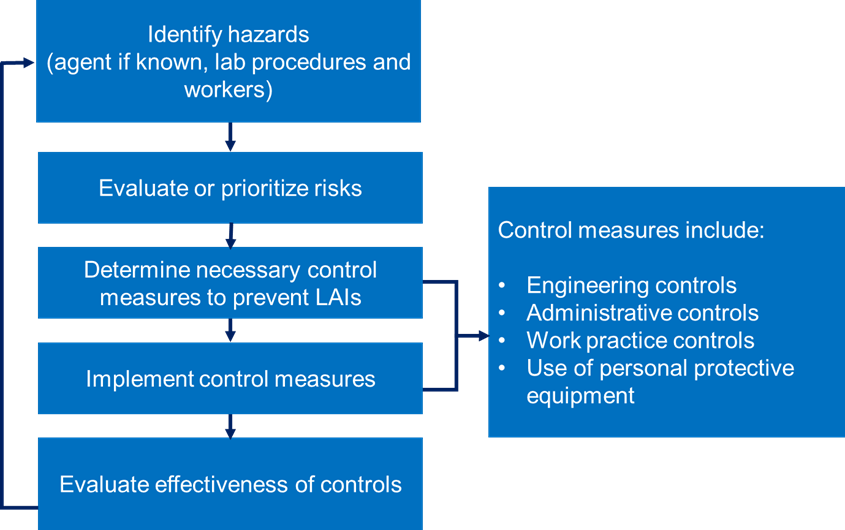
Figure: Risk assessment process for a biological hazard
Risk assessments for TB laboratories should consider:
- Bacterial load of materials (such as specimens and cultures)
- Viability of the bacilli
- Whether the material handled is prone to generate infectious aerosols during different procedures
- Laboratory’s workload and procedures; it is important that laboratories working with drug resistant strains should establish higher level precautions and biosafety measures
- Epidemiology of the disease and the patient population served by the laboratory
- Health/immune status of laboratory workers
- Experience and competency of laboratory staff in using good laboratory practices while handling infectious agents
- Equipment biosafety features required to prevent spill/breakage/aerosol generation when handling potentially infectious material
- Maintenance of equipment
- Biomedical waste management
Resources
Kindly provide your valuable feedback on the page to the link provided HERE
Biosafety in TB Laboratory: Risk Monitoring
ContentRisk monitoring is a regular inspection of the laboratory environment to monitor risks and control measures.
Stages of Risk Monitoring
1. Thorough investigation of lab incidents/lab accidents to understand the reason for such accidents
2. Review of corrective actions taken after the lab incident/lab accident
3. Implementation of preventive measures post-incident to prevent the future occurrence of such events
4. Documenting the process of risk monitoring
Importance of Risk Monitoring
Risk monitoring is important to ensure that selected and implemented biosafety measures are constantly improved.
Development of a new procedural risk assessment/ review of an existing one is required in the following situations:
- If there is an initiation of new work or there is a change in the work environment
- If there are some alterations to the workflow or volume of work done
- Construction of a new lab or modification in the lab structure
- Introduction of new equipment into the lab
- Alterations in staffing arrangements
- Alterations in standard operating procedures or working practices
- Major incidents in the laboratory like a major spill
- Evidence of, or suspicion of, any staff acquiring a Laboratory Acquired Infection
- Emergency responses and contingency planning requirements
Resources
Kindly provide your valuable feedback on the page to the link provided HERE
Biosafety in TB Laboratory: Factors to be Considered in TB Laboratory Risk Assessment
ContentStaff working with patient specimens and live mycobacterial cultures must operate under appropriate biosafety conditions with adequate infection control measures in place, including staff health checks.
For the safety of personnel working in TB labs, the following factors must be considered to avoid the risk associated with pathogenicity and transmission of TB bacilli.
The 4 key factors that must be considered during risk assessment of TB labs include:
1. The organism i.e., M. tuberculosis: Different strains of TB carry different levels of individual and community hazards. The following should be considered:
- Route of transmission (Inhalation of aerosols)
- TB epidemiology and patient population served by the laboratory (drug-sensitive TB, drug-resistant TB, non-TB patient proportions): MDR and XDR strains carry higher risks and cause greater harm to the infected individual as treatments may be limited or less effective.
2. Specimen Handling: Procedures involving generation of aerosols are more hazardous. The following should be considered:
- Bacterial load in the environment
- Viability of bacilli in sputum specimens and cultures
- Manipulations (centrifugation, vortexing, and vigorous shaking) that are likely to generate infectious aerosols
3. Staff and Workload: Individuals in the laboratory differ in their susceptibility to TB, higher-risk individuals may be less experienced, over-worked or have an underlying health condition. The following should be considered:
Level of experience and competency of the laboratory staff:
- Training status of staff to perform laboratory procedures
- Staff’s competency procedures in handling potentially infectious material and response to emergencies
- Ability of the staff to use biosafety equipment properly
- Review of the technical proficiency of staff
- Staff proficiency in using aseptic techniques and Biological Safety Cabinets (BSCs)
- Staff willingness to accept responsibility for protecting themselves and co-workers
Health status of staff: Higher-risk groups include:
- Individuals with reduced immunity which may be caused by certain medications
- HIV-infection or pregnancy may increase the risk of becoming infected with TB
- Diabetic individuals
4. Location of the Laboratory: TB Laboratories are generally located away from the main health facilities in separate building with unrestricted entrance to the building. If the laboratory cannot be located in a separate, dedicated building, separation may be achieved by placing the laboratory at the blind end of a corridor. TB containment laboratories should have access only to authorized staff through an anteroom.
Resources
Kindly provide your valuable feedback on the page to the link provided HERE
Fullscreen