Shorter oral Bedaquiline-containing MDR/RR-TB regimen
ContentBased on the World Health Organization (WHO) treatment guidelines, 2020 recommendations, the National TB Elimination Programme (NTEP) have decided to transition from the current shorter injectable-containing Multi-drug Resistant (MDR)/ Rifampicin-resistant TB (RR-TB) regimen to the shorter oral bedaquiline-containing MDR/RR-TB regimen in the year 2021.
Salient Features of the Shorter Oral Bedaquiline-containing MDR/RR-TB Regimen
- This regimen will be used in patients >5 years of age weighing 15 kg or more.
- The regimen will be expanded in a phased manner starting with selected states to gain programmatic experience to guide future expansion within 2021.
- The regimen consists of an initial phase of 4 months that may be extended up to 6 months and a continuation phase of 5 months, giving a total duration of 9-11 months. Bdq is used for a duration of 6 months.
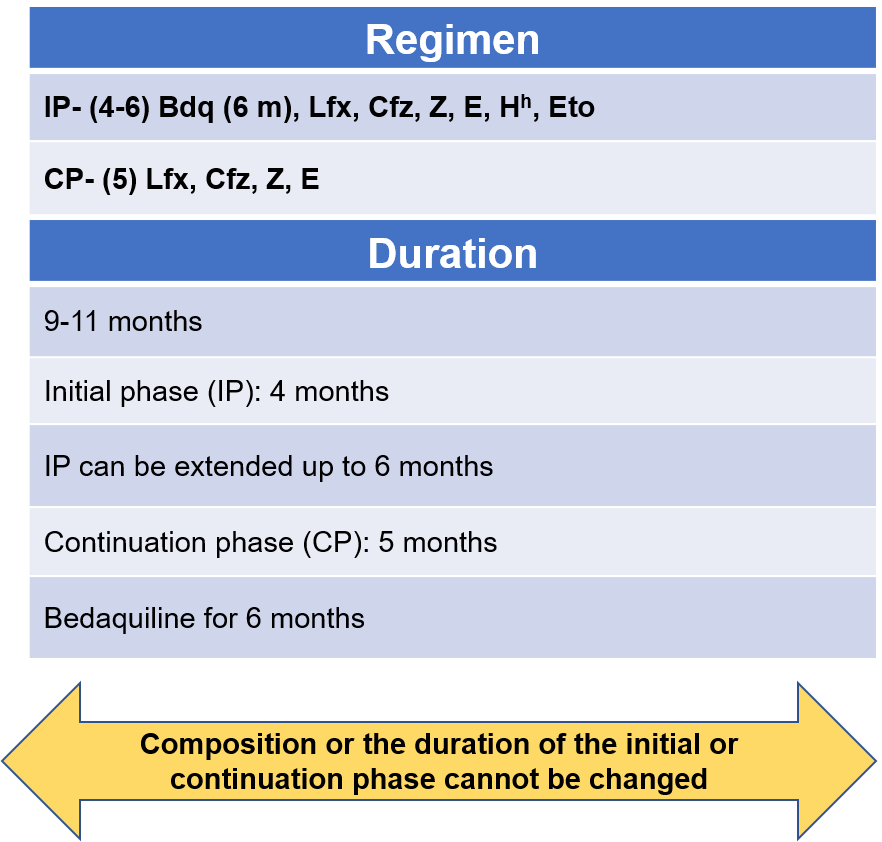
Figure 1: Regimen and duration of shorter oral Bdq-containing MDR/RR-TB regimen
Abbr: Bdq - Bedaquiline, Lfx- Levofloxacin, Cfz- Clofazamine, Z- Pyrazinamide, E- Ethambutol, Hh- High-dose Isoniazid, Eto- Ethionamide
Points to Note
- From the start to the end of 4 months these drugs are used: Bedaquiline, Levofloxacine, Clofazamine, Pyrazinamide, Ethambutol, high-dose Isoniazid, Ethionamide
- From the start of 5 months to the end of 6 months (If IP not extended): Bdq, Lfx, Cfz, Z, E
- From the start of 7 months to the end of 9 months: Lfx, Cfz, Z, E
If the IP is extended up to 6 months, then all 3 drugs Bdq, Hh and Eto are stopped together
Resources
- Guidelines for Programmatic Management of Drug-resistant Tuberculosis in India, March 2021.
- WHO Consolidated Guidelines on Tuberculosis, Module 4 - Treatment: Drug Resistant TB Treatment.
Kindly provide your valuable feedback on the page to the link provided HERE
Shorter Oral Bedaquiline-containing MDR/RR-TB Regimen: Pre-treatment Evaluation [PTE]
ContentThere are two main evaluations that must be conducted during pre-treatment evaluation to initiate patients on a shorter oral bedaquiline-containing MDR/RR-TB regimen. These evaluations are:
Clinical Evaluation
- History and physical examination
- Height check
- Weight check
- Psychiatric evaluation, if required
Laboratory-based Evaluation
- Random Blood Sugar (RBS)
- HIV testing following counselling
- Complete blood count (Haemoglobin (Hb), Total Leucocyte Count (TLC), Differential Leucocyte Count (DLC), platelet count)
- Liver function tests (including serum proteins Thyroid-stimulating Hormone (TSH) levels)
- Urine examination – routine and microscopic
- Serum electrolytes (Sodium (Na), Potassium (K), Magnesium (Mg), Calcium (Ca))
- Urine pregnancy test (in women of reproductive age)
- Chest X-ray
- Electrocardiogram (ECG)
Resources
- Guidelines for Programmatic Management of Drug-resistant Tuberculosis in India, March 2021.
- WHO Consolidated Guidelines on Tuberculosis: Module 4-Treatment: Drug-resistant TB Treatment, 2020.
Kindly provide your valuable feedback on the page to the link provided HERE
Shorter Oral Bedaquiline-containing MDR/RR-TB Regimen: Treatment Extension
ContentThe total duration of treatment in this regimen is 9-11 months with Intensive Phase (IP) at least 4 months and Continuation Phase (CP) for 5 months. Treatment extension of IP is done up to 2 months based on follow-up results and is indicated in the algorithm presented in the figure below.
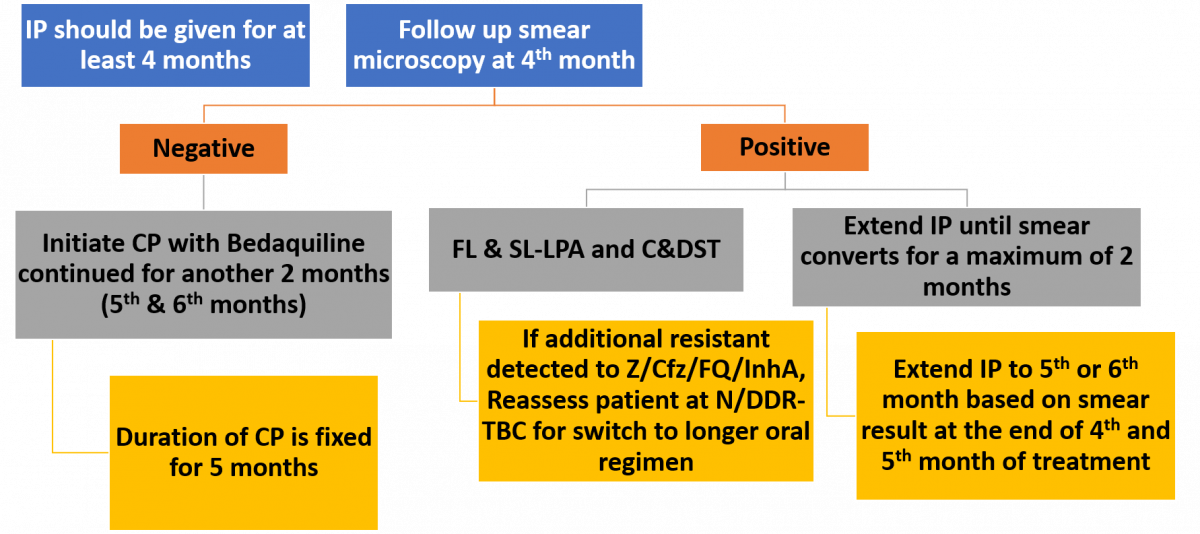
Figure: Treatment Extension/ Regimen Change Based on Follow up Smear/ Culture/ DST Results
Abbr: FL & SL- LPA- First Line & Second Line- Line Probe Assay, C&DST- Culture and Drug Susceptibility Test, Z- Pyrazinamide, Cfz- Clofazimine, FQ- Fluoroquinolone, N/DDR-TBC- Nodal/ District DR-TB Centre
Resources
- Guidelines for Programmatic Management of Tuberculosis in India, 2021.
- WHO Consolidated Guidelines on Tuberculosis: Module 4 – Treatment: Drug-resistant TB Treatment, 2020.
Shorter Oral Bedaquiline-containing MDR/RR-TB Regimen: Drug Dose Administration
ContentDrug dose administration for shorter/ longer oral Bedaquiline (Bdq)-containing Multidrug-resistant (MDR)/ Rifampicin-resistant (RR)-TB regimen depends on the factors described below.
- The dosage of second-line anti-TB drugs would vary as per the weight of the Drug-resistant TB (DR-TB) patients. There are five weight bands in adult patients (≥ 18 years): <16 kg, 16-29 kg, 30-45 kg, 46-70 kg and >70 kg. The weight bands of adult patients for Drug-sensitive TB (DS-TB) patients are different compared to DR-TB patients.
- All morning doses should be supervised by the treatment supporter via Directly Observed Treatment, Short-course (DOTS).
- In patients with drug intolerance, Cycloserine (Cs), Ethionamide (Eto) and Sodium (Na) Para Aminosalicylic Acid (PAS), can be given in two divided doses. Pyridoxine should be provided as part of the regimen till the end of treatment for all patients.
Change in Weight Bands during Treatment
- For adult DR-TB patients whose weight increases or decreases by 5 kg or more compared to baseline weight and crosses the current weight band during the course of the treatment, the weight band must be changed at the time of issuing next month's box to the treatment supporter of the patient.
- For paediatric patients, the drug dosage should be adjusted immediately once the weight of the patient crosses the range of the weight band. Counsel the patient about the change in dose.
Key Considerations for Newer Drugs
- Avoid milk-containing products with drugs: The calcium in the milk can decrease the absorption of Fluoroquinolones (FQs).
- Avoid large fatty meals: Fat impairs the absorption of anti-TB drugs (Cs, Isoniazid (H), etc.).
Resources
Shorter Oral Bedaquiline-containing MDR/RR-TB Regimen: Dosage of Drugs for Adults
ContentIn adults, the dosage of drugs for a shorter oral Bedaquiline (Bdq) - containing Multi-drug Resistant (MDR)/ Rifampicin-resistant Tuberculosis (RR-TB) regimen, varies according to their weight.
The table below provides drug dosages for adult patients, according to their weight bands, in a shorter oral Bedaquiline-containing MDR/RR-TB regimen.
Table: Weight-bandwise Dosages for Adult Patients on Shorter Oral Bdq-containing MDR/ RR-TB Regimen Sr No
Drugs
16-29 kg
30-45 kg
46-70 kg
>70 kg
1
High dose H (Hh)
300 mg
600 mg
900 mg
900 mg
2
Ethambutol (E)
400 mg
800 mg
1200 mg
1600 mg
3
Pyrazinamide (Z)
750 mg
1250 mg
1750 mg
2000 mg
4
Levofloxacin (Lfx)
250 mg
750 mg
1000 mg
1000 mg
5
Bedaquiline (Bdq)
Week 0–2: Bdq 400 mg daily
Week 3–24: Bdq 200 mg 3 times per week
6
Clofazimine (Cfz)
50 mg
100 mg
100 mg
200 mg
7
Ethionamide (Eto)*
375 mg
500 mg
750 mg
1000 mg
8
Pyridoxine (Pdx)
50 mg
100 mg
100 mg
100 mg
*Drugs can be given in divided doses in a day in the event of intolerance. Resources
- Guidelines for Programmatic Management of Drug-resistant Tuberculosis in India, March 2021.
- The Use of Bedaquiline in the Treatment of Multidrug-resistant Tuberculosis: Interim Policy Guidance, 2013.
Kindly provide your valuable feedback on the page to the link provided HERE
Possible Adverse Events Due to Drugs in Shorter Oral Bedaquiline-containing MDR/RR-TB Regimen
ContentDrugs that are part of the shorter Bedaquiline (Bdq)-containing regimen have some typical side effects which need close monitoring of Drug Resistant-TB (DR-TB) patients while providing the treatment.
Table: Possible adverse events due to drugs in the shorter oral Bedaquiline-containing MDR/RR-TB regimen; Source: Guidelines for PMDT in India, 2021.
Adverse Drug Events Causative Drugs QT prolongation Bdq, Fluoroquinolone (FQ), Clofazimine (Cfz) Rash, allergic reaction and anaphylaxis Any drug Gastrointestinal symptoms Ethionamide (Eto), Pyrazinamid (Z), Ethambutol (E), Bdq, Cfz, FQs, Isoniazid(H) Diarrhoea and/or flatulence Eto Hepatitis Z, H, Eto, Bdq Giddiness Eto, FQ, Z Hypothyroidism Eto Arthralgia Z, FQ, Bdq Peripheral neuropathy H, FQ, rarely Eto, E Headache Bdq Depression FQ, H, Eto Psychotic symptoms Isoniazid (H), FQ Suicidal ideation H, Eto Seizures H, FQ Tendonitis and tendon rupture FQ Vestibular toxicity (tinnitus and dizziness) FQs, H, Eto Optic neuritis E, Lzd, Eto, Cfz, H Metallic taste Eto, FQs Gynaecomastia Eto Alopecia H, Eto Superficial fungal Infection and thrush FQ Dysglycaemia and Hyperglycaemia Eto Resources
Kindly provide your valuable feedback on the page to the link provided HERE
Follow-up Evaluation of Patients on Shorter Oral Bedaquiline-containing MDR/RR-TB Regimen
ContentApart from clinical evaluation, the patients need to be closely assessed by various laboratory parameters to monitor the improvement on treatment, drug-induced adverse events or co-morbidities to enable timely interventions to address these and improve the probability of treatment success, survival and quality of life.
Table: Laboratory evaluations and follow-up schedule for patients on shorter oral Bdq containing Multidrug-resistant (MDR)/ Rifampicin-resistant (RR) TB regimen; Source: Guidelines for PMDT in India 2021, p54.
EVALUATION TEST
FOLLOW-UP SCHEDULE
Clinical + Weight (Wt.)
Monthly in Intensive Phase (IP) or extended IP if the previous month shows Smear-positive (S+ve), quarterly in Continuation Phase (CP)
Smear Microscopy (SM)
- Monthly from 3rd month onwards till end of IP
- Monthly in extended IP only if the previous month S+ve (IP extension up to a maximum of 6 months)
- Conduct SM within 7 days, if the smear at 6 months is positive to rapidly ascertain bacteriological conversion/ reversion.
Culture
- At the end of month 3, end of month 6 and/or end of treatment
- If the culture result of month 6 is positive, collect one repeat sample immediately to rapidly ascertain the bacteriological conversion/ reversion.
- If the repeat sample is culture-negative, then end of treatment specimen collection should be done.
Drug Susceptibility Testing
(DST)
First-line and Second-line Line Probe Assay (FL-SL LPA) (Levofloxacin (Lfx), Moxifloxacin (Mfx), Ethionamide (Eto)) and Liquid Culture and Drug Susceptibility Testing (LC&DST) (Pyrazinamide (Z), Bedaquiline (Bdq)*, Clofazimine (Cfz)*, Mfx, Linezolid (Lzd), Delamanid (Dlm)*) if any of the following:
- Culture +ve (end of month 3 or later and end of treatment)
- Smear +ve at end of IP, end of extended IP and end of treatment
Urine Pregnancy Test (UPT)
As and when clinically indicated
Complete Blood Count (CBC)
As and when clinically indicated
Thyroid Stimulating Hormone (TSH) and Liver Function Test (LFT)#
At end of IP, then as and when clinically indicated
Chest X-ray (CXR)
At end of IP, then as and when clinically indicated, end of treatment
Electrocardiogram (ECG)$
At 2 weeks, then monthly in the first 6 months, then as and when clinically indicated
Serum Electrolytes
(Na, K, Mg, Ca)
As and when indicated and in case of any QTcF prolongation
Specialist consultation
As and when clinically indicated
Colour vision test
Once in two months (in children)
# HBsAG and other viral markers (Hepatitis A, C & E) to be done in case of Jaundice.
$ In case of baseline ECG abnormality or QTcF ≥450ms with a shorter oral Bedaquiline-containing MDR/RR-TB regimen that contains Bdq and Cfz, ECG must be done on daily basis for initial 3 days or as suggested by a cardiologist. Repeat ECG with long II lead after an hour to reconfirm abnormal ECG.
DST whenever available.
Important Points
- The most important evidence of response to DR-TB treatment is the conversion of sputum smear and culture to negative.
- If no additional resistance is detected on follow-up after 3rd month, the IP will be extended on monthly basis up to a maximum of 6 months.
If bacteriological reversion is ascertained or if any resistance is detected by FL-LPA or SL-LPA or if found to be smear/ culture positive at the end of 6 months or later, the patient will be declared as ‘treatment failed’.
- The patient is then re-evaluated for a longer oral Multi (M)/ Extensively Drug-resistant TB (XDR-TB) regimen with appropriate modification if required.
- A patient once treated with the shorter oral Bedaquiline-containing MDR/RR-TB regimen for more than one month will never be reinitiated on it again.
Resources
- Guidelines for Programmatic Management of Drug-resistant Tuberculosis in India, March 2021.
- WHO Consolidated Guidelines on Tuberculosis: Module 4 - Treatment: Drug-resistant TB Treatment, 2020.
Kindly provide your valuable feedback on the page to the link provided HERE
Fullscreen