Requirements for Packaging a biological Specimen
ContentPeripheral Laboratories in the NTEP need to send biological samples (such as sputum) to nearby Culture and Drug Susceptibility Testing (CDST) laboratories for advanced testing (eg Drug Resistance Testing). The samples need to be safely packaged and transported such that there is no spillage or contamination.
The items required for safe packaging biological specimens are:
- Falcon tubes
- Three-layer packing materials like thermocol box
- Ice gel pack (pre-frozen at -20oC for 48 hours)
- Request for CDST forms
- Polythene bags
- Tissue paper roll for absorbent packing
- Parafilm tapes
- Brown tape for packing the thermocol box
- Permanent marker pen
- Labels with 'To' and 'From' address and blank labels for sample details
- Biohazard sticker
- Scissors
- Spirit swab
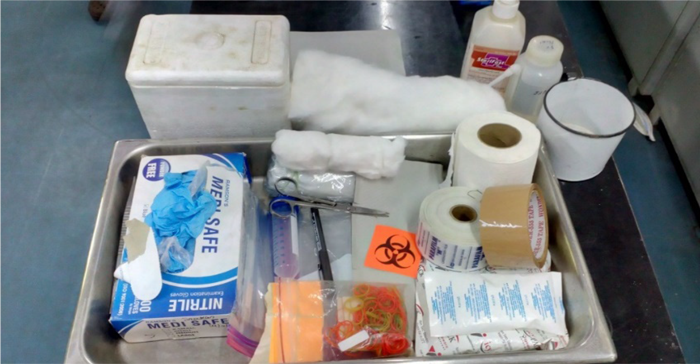
Figure: Items needed for packaging of biological specimens for CDST laboratory; Source: Guidelines for Programmatic Management of Drug Resistant TB in India, 2021
Video fileResources
Specimen Carriers
ContentTechnical specifications for TB diagnosis specimen carriers are as follows:
Thermocol Box:
- Thickness of box - 2.5 cm
- Outer dimensions: Length - 18.5 cm, breadth - 13 cm, height - 12 cm (without lid), height - 14 cm (with lid); Inner dimensions: Length - 14.5 cm, breadth - 8 cm, height - 12 cm (without lid), height - 13 cm (with inner part of lid).
Gel Pack:
- Number of gel packs required: 2
- Weight of fully packed consignment box: 400 grams
- Gel packs maintain a temperature between 12-20ºC for up to approximately 48 hours in tightly packed thermocol boxes (average outside temperature 35ºC)
- Conditioning in the deep freezer (temperature between -20 to -15ºC) for a minimum 48 hours to a maximum 72 hours before use, is required.
This is a one-time use carrier since the thermocol boxes and the gel packs are not reused.

Figure: TB Diagnostic Specimen Carrier - Thermocol Box with Gel Packs; Source: NTEP Guidelines for Programmatic Management of Drug Resistant TB in India, 2021
Resources
- Training Module (1-4) for Program Managers and Medical Officer, NTEP, MoHFW, 2020.
- Guidelines for Programmatic Management of Drug Resistant TB in India, 2021.
Kindly provide your valuable feedback on the page to the link provided HERE
SOP for Packaging Specimens during the Transportation of Biological Specimens for TB Diagnosis
ContentStandard Operating Procedure (SOP) for Packaging Specimen during the transportation of biological specimens for TB diagnosis, also known as Triple Layer Packaging, is as follows:
The Lab Technicians (LTs) at the Designated Microscopy Centres (DMCs) should be trained to carefully pack the sputum samples in the cold box to avoid spillage of the samples. Before packing, personal protection measures such as wearing hand gloves (double gloves preferred), goggles and masks are to be followed by the LTs to prevent contracting the infection.
Table: Steps in Standard Operating Procedure (SOP) for Packaging Specimen during Transportation for TB Diagnosis
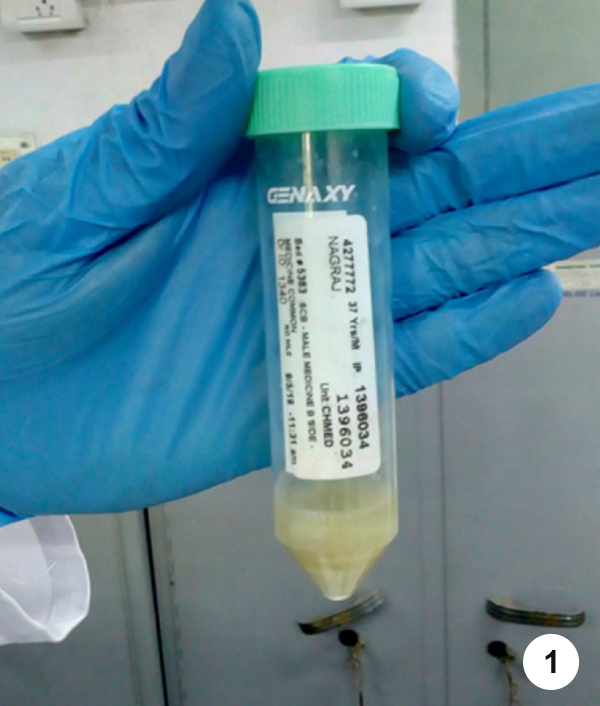
- Step 1. Make sure that the specimen collection tube is tightly closed after the sample has been collected from the patient.

- Step 2. Wipe the outer surface of the 50 ml conical tube with 5% phenol followed by absorbent tissues and allow it to air dry.

- Step 3. Write the patient details on the opaque area (white area) of the specimen collection tube using a permanent marker pen, clearly in capital letters.
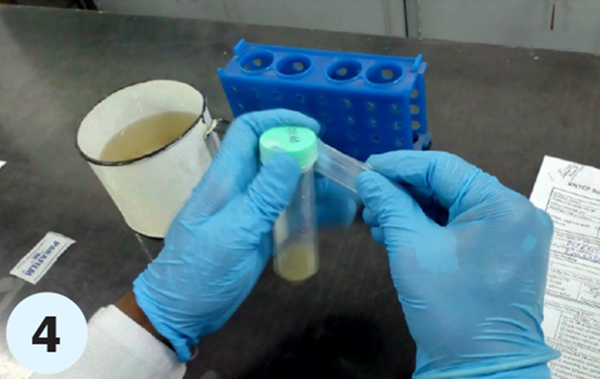
- Step 4. Cut the parafilm strip and wrap one of the strips at the joint between the cap and the neck of the specimen collection tube such that a secured seal is formed. (Primary receptacle/ package

- Step 5. Open the absorbent cotton roll and spread it out on the workbench; separate the cotton into two equal layers. Roll the specimen collection tube containing the sample tightly in the absorbent cotton such that the tube is covered completely.
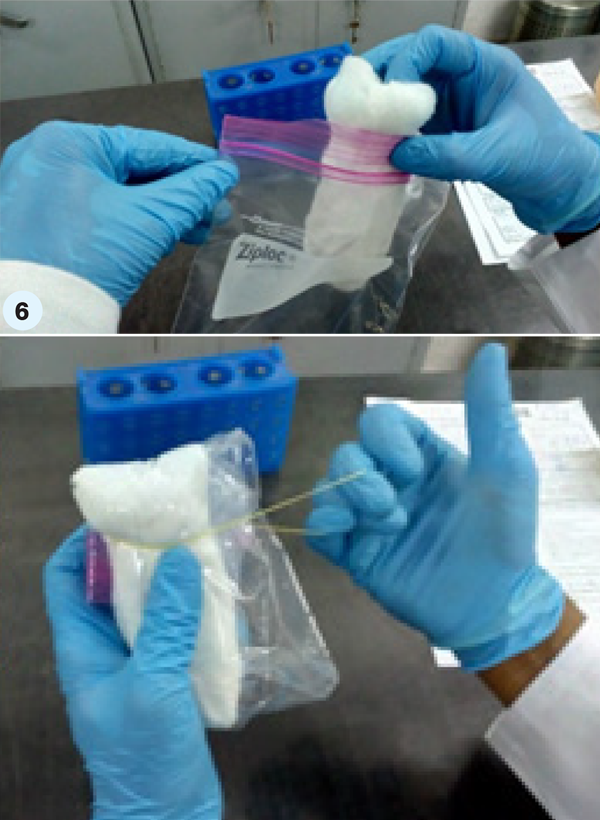
- Step 6. Put this roll containing the specimen collection tube into the ziplock pouch. Roll the whole into a tight bundle, ensuring that there is no air in the pouch. This bundle should be secured with rubber bands. (Secondary receptacle/ package)


- Step 7. Repeat steps 5–7 for the second sample of the patient.

- Step 8. Insert the Test Request form printed from Nikshay into the ziplock pouch after ensuring that the details on the form and the sample tubes match, with the writing facing outside (details visible through the package). Seal the ziplock on the pouch.

- Step 9. Place the cooled gel packs into the thermocol box, place the sample tubes packed in ziplock pouches on the frozen gel packs (frozen for 48 hours at -40°C) and also keep the pouch containing the Test Request form printed from Nikshay on top. Stick the BIOHAZARD sign over the lid and “To and From” stickers on the exterior of the thermocol box or box used to pack the specimen. Close the lid of the box and wrap it tightly with brown duct tape. (Tertiary receptacle/ package)

- Step 10. Complete the ‘From’ and ‘To’ addresses on the stickers, using a permanent marker pen.
The LT of the DMC should promptly inform the sample transport agency like a courier/ speed post service, or a human carrier to collect and transport the samples.
Video fileResources
- Training Module (1-4) for Program Managers and Medical Officers, NTEP, MoHFW, 2020.
- Guidelines for Programmatic Management of Drug-resistant TB in India, 2021.
Kindly provide your valuable feedback on the page to the link provided HERE
Technical Specifications for Labelling Specimen Transport Carriers
ContentThese are the technical specifications for labelling specimen transport carriers:
- Specimen containers need to be labelled legibly with details such as the patients’ name, date and time of specimen collection, TB detection centre/ District Tuberculosis Centre (DTC), lab no., specimen A or B.


Figure: Details to be filled on the specimen containers

- Dispatch list with details of each specimen transported and the request form for examination of biological specimen for each specimen should be put in an envelope and attached to the outside box.
- As per the national guidelines for biomedical waste management, the containers used for transporting specimen samples to the National TB Elimination Programme (NTEP) - certified laboratory should be labelled with a “BIOHAZARD” sticker.

- Other than the Biohazard sticker it is mandatory to add 'To' and 'From' stickers on the specimen transportation carrier and fill in the necessary details.
- Specimen transport carriers should be labelled legibly with all the necessary details as listed above.
- A specimen may be rejected at the receiving laboratory if the specimen transport carriers are unlabelled or mislabelled.
Resources
Kindly provide your valuable feedback on the page to the link provided HERE
Dispatching the sample to a C&DST Lab
ContentFor presumptive Drug-resistant TB (DR-TB) patients’, the health facility staff arranges for specimen collection from patient, packs samples as per the Standard Operating Procedure (SOP) for triple layer packaging and dispatches it for transportation in cool chain to the linked Culture and Drug Susceptibility Testing (C&DST) laboratory.
All necessary materials for specimen collection and modality for transportation need to be made available/ arranged at the Designated Microscopy Centre (DMC)/ Peripheral Health Institute (PHI) by the District TB Officer (DTO).
Steps in sample dispatch include:
- Add test details in Nikshay (Figure) to generate test requests (Test ID) for the patient’s episode ID in Nikshay.
- This will enable instant online intimation about the upcoming specimen to the C&DST laboratory.
- Complete test request form for biological specimen by adding:
- Patient information
- Details on name and type of referring facility
- Health establishment ID
- Reason for testing and test requested
- Patient ID and Test ID
- Put the appropriate address of receiving C&DST laboratory and address of health facility form where samples are dispatched on the transportation box.
- Lab Technician (LT) of the health facility should promptly inform the transport agency (courier/ speed post service) or human volunteers to collect and transport the samples.

Figure: Adding Test Details in Nikshay for Diagnosis of DR-TB; Source: Nikshay Diagnostics Training Content.
Resources
- Training Modules (1-4) for Programme Managers and Medical Officers, NTEP, 2020.
- Guidelines for PMDT in India, 2021.
- Nikshay Zendesk, Nikshay Knowledge Base, Diagnostics.
Assessment
Question
Answer 1
Answer 2
Answer 3
Answer 4
Correct answer
Correct explanation
Page id
Part of Pre-test
Part of Post-test
Test details are added in Nikshay to generate test requests (Test ID) for the patient’s episode ID in Nikshay.
True
False
1
Test details are added in Nikshay to generate test requests (Test ID) for the patient’s episode ID in Nikshay.
Yes
Yes
- Add test details in Nikshay (Figure) to generate test requests (Test ID) for the patient’s episode ID in Nikshay.
Fullscreen

