TB Notification Register
ContentVideo fileVideo: TB Notification Register
Tuberculosis Laboratory Register
ContentThe Tuberculosis (TB) Laboratory Register is a paper-based recording register kept in all National TB Elimination Programme (NTEP) laboratories for recording details of diagnostic services offered to TB patients referred from both private and public health facilities.
The register is maintained in the Designated Microscopy Centre (DMC). It is the only register used for recording the details of specimen smear examinations. The Laboratory Technician (LT) is responsible for maintaining and updating the laboratory register.
There are two portions in the TB lab register and the table below shows the details captured in each portion.
Table: Pages and Information Covered in the TB Laboratory Register; Source: NTEP Training Module 2 for Programme Managers & Medical Officers
PORTION
DETAILS CONTAINED
Left-hand Portion
Lab serial number assigned by the LT
Date of the sample collection
Patient details such as name, sex, age, address, and contact number
Information on if the patient is from a key population
Referring health facility details
Reasons for examination
Right-hand Portion
Details on the type of specimen
Visual appearance results along with dates
Comorbidity status (HIV and Diabetes)
Details of drug susceptibility testing
Nikshay ID / Notification
Treatment initiation details
The last two columns are for the LT’s signature and remarks by the LT or supervisor
Important Points to Note
- Duplicate registers should not be maintained.
- LTs should ensure that the correct laboratory serial number is recorded.
- Laboratory serial number is given to the patient and not to the sample. A new number should be assigned to every presumptive TB case whose sputum is to be examined.
- All smear-positive (including scanty) results should be recorded in red ink.
- No over writing or manipulation in already entered data should be done.
The figure below shows the left-hand portion of the TB lab register. Click here to access the full form in the NTEP Training Module 2 for Programme Managers & Medical Officers, p. 219.
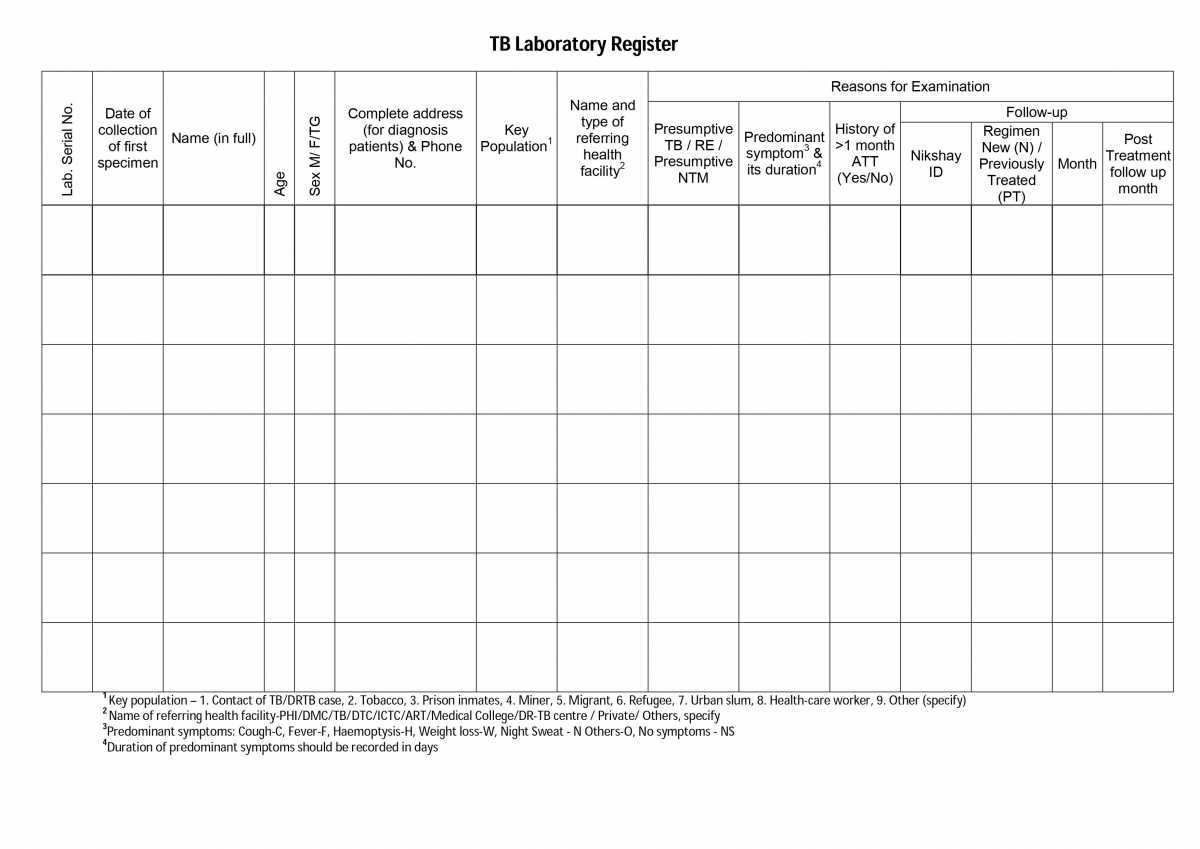
Figure: First Page of the TB Laboratory Register; Source: NTEP Training Modules 1-4 for Programme Managers & Medical Officers, p. 219
Resources
Kindly provide your valuable feedback on the page to the link provided HERE
Reporting the performance of a DMC
ContentThe monthly abstract is a recording format filled and printed in the Designated Microscopy Centre (DMC) Tuberculosis (TB) laboratory register. Every month after the last day, the laboratory technician (LT) summarizes the information on sputum smear examinations done during that month, using the monthly abstract format.
Additionally, on-site evaluation of the technical practices in the National TB Elimination Programme (NTEP) laboratories is performed by a properly trained Senior Tuberculosis Laboratory Supervisor (STLS). The major findings of this supervision visit are recorded as part of the monthly abstract in the TB laboratory register.
Important Points to Note
- The STLS should write the monthly supervisory abstract after the last entry of the month.
- A new page is used every month for recording the sputum examinations undertaken.
- The correctness of the information is very important as a small mistake at a particular DMC can change the overall figures of a particular district.
The figure below shows the TB lab monthly abstract format.

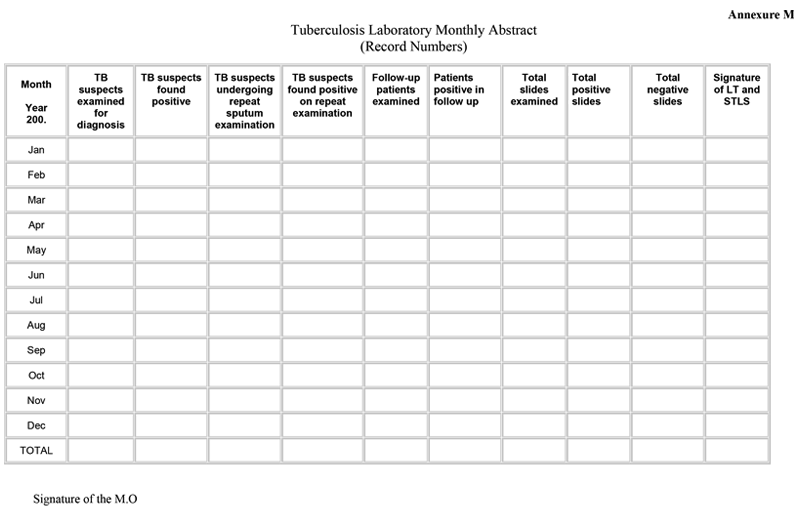
Figure: TB Lab Monthly Abstract Format; Source: Guidelines for Quality Assurance of Smear Microscopy for Diagnosing Tuberculosis; p85.
Resources
- Guidelines for Quality Assurance of Smear Microscopy for Diagnosing Tuberculosis, NTEP, 2005.
- NTEP Training Modules 1-4 for Programme Managers & Medical Officers, 2020.
Kindly provide your valuable feedback on the page to the link provided HERE
Fullscreen
