Cartridge Based Nucleic Acid Amplification Test [CBNAAT]
ContentCartridge Based Nucleic Acid Amplification Test (CBNAAT) is a rapid molecular diagnostic test. It is used for diagnosis of Tuberculosis (TB) and Rif-resistant Tuberculosis (RR-TB) in NTEP. Results are obtained from unprocessed sputum samples in about 2hours which helps in early detection and treatment of TB patients.
India has vast number of CBNAAT laboratories which are utilized for TB/RR-TB detection and Universal Drug Susceptibility Testing (UDST) under the National TB Elimination Program (NTEP).

Figure: CBNAAT Cartridge and Machine in Use (Image courtesy: USAID supported Challenge TB Project)
The CB-NAAT system detects DNA sequences specific for Mycobacterium tuberculosis complex and rifampicin resistance by Polymerase Chain Reaction (PCR). It concentrates Mycobacterium tuberculosis bacilli from sputum samples, isolates genomic material from the captured bacteria by sonication and subsequently amplifies the genomic DNA by PCR. The process identifies clinically relevant rifampicin resistance-inducing mutations in the RNA polymerase beta (rpoB) gene in the Mycobacterium tuberculosis genome in a real-time format using fluorescent probes called molecular beacons.
Video fileVideo: Cartridge-Based Nucleic Acid Amplification Test [CBNAAT] - GeneXpert Technology
Resources
- Training Module (1-4) for Program Managers and Medical Officers, NTEP, MoHFW, 2020.
- India TB Report 2021, National TB Elimination Program (NTEP), MoHFW, 2021.
Assessment Questions
Question
Answer 1
Answer 2
Answer3
Answer 4
Correct Answer
Correct explanation
Part of pre-test
Part of post-test
Under NTEP, CBNAAT is offered upfront for which of these categories?
PLHIV
Paediatric presumptive TB
Presumptive DR-TB
All of the above
4
Under NTEP, CBNAAT is recommended upfront for People living with HIV, Paediatric Presumptive TB patients, Presumptive DR-TB patients and patients notified from the Private sector.
Yes
Yes
CBNAAT requires the processing of sputum samples before testing
True
False
2
Results are obtained from unprocessed sputum samples in about 2hours from a CBNAAT machine
Yes
Yes
Structure and Function of the CBNAAT Module
ContentThe CBNAAT module contains components that enable automated sample processing in the cartridge, and filling of the tube with the sample-reagent mixture for Polymerase Chain Reaction (PCR), followed by PCR amplification and detection.
The structure of the CBNAAT module is described below and shown in Figure 1:
- Syringe pump drive or plunger motor: Dispenses fluids into the different cartridge chambers
- I-CORE module: Performs PCR amplification and detection
- A cartridge loading and unloading mechanism ensures the proper movement of the cartridge in the instrument
- Reaction/PCR tube: Enables rapid thermal cycling and optical excitation and detection of the tube contents. The reaction tube is automatically inserted into the I-CORE module (hardware for PCR amplification and fluorescence detection) when the cartridge is loaded into the instrument.
- Valve drive: Rotates the cartridge valves to align the chambers with the syringe
- Ultrasonic horn: Lyses the sample

Figure 1: Structure of the CBNAAT module
The interior region of the cartridge insertion site also called cartridge bay and is shown in Figure 2.

Figure 2: Interior of the Cartridge Bay
Mode of Operation of the CBNAAT Module
- The Intelligent Cooling/Heating Optical Reaction (I-CORE) module is the hardware component, within each instrument module, that performs PCR amplification and, fluorescence detection.
- As part of the cartridge load process, the PCR tube is inserted into the ICORE module.
- As the test starts, the sample and reagent mixture are pushed from the cartridge, into the PCR tube.
- During the amplification process, the ICORE heater heats up and the fan cools down the reaction tube contents.
Within the I-CORE, there is an optical system composed of two blocks: A six colour excitor and detector block excite the dye molecules, and detect the fluorescence emitted.
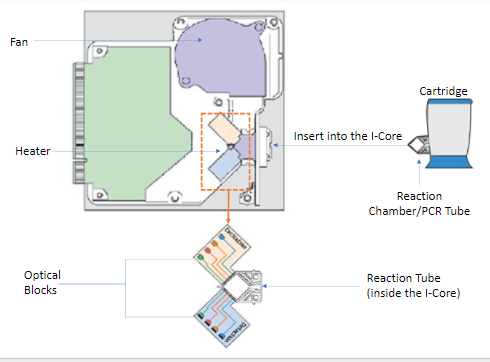
Figure 3: Mode of operation of the CBNAAT module
Video fileResources
Kindly provide your valuable feedback on the page to the link provided HERE
The CBNAAT Cartridge
Content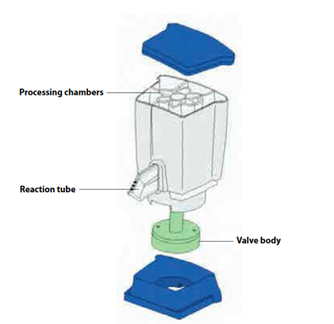
The disposable, single-use CBNAAT cartridge is a closed system unit to hold the samples and reagents. Inside of a CBNAAT Cartridge (CBNAAT operator manual). Each cartridge consists of the following components:
- Processing chambers: Hold samples, reagents, processed samples, and waste solutions. One chamber is designated as an air chamber to equilibrate pressures within the cartridge.
- Valve body: Rotates and allows fluid to move to different cartridge chambers and to the reaction tube. Within the valve body, the specimen is isolated, PCR inhibitors are removed, and the sample is mixed with PCR reagents and moved into the integrated reaction tube.
- Reaction tube: Enables rapid thermal cycling and optical excitation and detection of the tube contents. The reaction tube is automatically inserted into the I-CORE module (hardware for PCR amplification and fluorescence detection) when the cartridge is loaded into the instrument.
Video fileResources
Consumables required at CBNAAT Lab
ContentThe consumables required at a Cartridge-based Nucleic Acid Amplification Testing (CBNAAT) laboratory include the following:
CBNAAT/ GeneXpert Dx System consisting of CBNAAT machine preloaded with assay software, Computer and the Barcode reader
- CBNAAT assay kit (Figure) consisting of:
- CBNAAT cartridges: Kit contains 10 or 50 individually packed cartridges.
- CBNAAT reagent: 8 ml volume pack per cartridge. The sample reagent solution is clear but may range from colourless to golden yellow.
- Sterile pipette: Individually packed, disposable transfer pipettes, one per each test, with a single mark for the minimum volume of sample transfer to each cartridge.
- CD containing the Assay Definition File.

Figure: Contents of CBNAAT Assay Kit; Source: GLI Training Package for CBNAAT.
- Sputum containers
- Personal protective equipment:
- Laboratory coats
- Disposable gloves
- Disinfectants
- 1% Sodium hypochlorite solution
- 5% Phenol
- 70% Ethanol
-
Power stabiliser (UPS) for uninterrupted power supply to perform CBNAAT assay.
Resources
Assessment
Question Answer 1 Answer 2 Answer 3 Answer 4 Correct answer Correct explanation Page id Part of Pre-test Part of Post-test What are the components of the CBNAAT assay kit? CBNAAT cartridges CBNAAT reagent Sterile pipette All the 3 4 CBNAAT assay kit includes CBNAAT cartridges, CBNAAT reagent and sterile pipette. Yes Yes - CBNAAT assay kit (Figure) consisting of:
CBNAAT Testing Process Overview
ContentCartridge-based Nucleic Acid Amplification Test (CBNAAT) is used to detect Mycobacteria tuberculosis and rifampicin-resistance using GeneXpert IV Dx system and the Xpert MTB/ RIF cartridge.
The CBNAAT system integrates and automates sample processing, nucleic acid amplification, and detection of Mycobacteria tuberculosis and rifampicin resistance.
The system utilises the use of single-use disposable CBNAAT cartridges that hold the Polymerase Chain Reaction (PCR) reagents and hosts the PCR process.
The process involves the following steps:
- Sample processing: Specimens are processed by adding the sample reagent at 2:1 (v/v) ratio to the sputum sample, mixing and incubation for 15 minutes at room temperature. The sputum sample get liquified after the processing step.
- Loading sample into cartridge: Liquefied sample is added to the cartridge using a transfer pipette.
- Setting up the machine to run the test: After switching on the system, “Create Test” (Figure A) is clicked in the GeneXpert Dx system window, test details are added, the barcode label of the cartridge is scanned (Figure B) and “Start Test” (Figure C) is clicked to initiate testing.

Figure: Setting the machine to run the assay by creating test (A), adding test details, scanning barcode (B) to start test (C); Source: GLI Training Package for CBNAAT.
- Loading the cartridge: The instrument module door which displays the blinking green light is opened to load the cartridge. The door of module is closed firmly (an audible click sound should be heard). The green light stops blinking when the test starts.
- Obtaining the results: When the test is finished, the green light turns off. It takes around 1 hour 55 minutes to complete test run. On completion of test run, the result is generated automatically on the monitor.
- Ending the test run: When the system releases the door lock at the end of run, the module door is opened to remove the cartridge. The used cartridge is discarded.
Resources
Assessment
Question
Answer 1
Answer 2
Answer 3
Answer 4
Correct answer
Correct explanation
Page id
Part of Pre-test
Part of Post-test
Barcode is present on sterile pipette and CBNAAT cartridge.
True
False
2
Barcode is present on CBNAAT cartridge.
Yes
Yes
Other CBNAAT tests available
ContentIntroduction
Cartridge-based Nucleic Acid Amplification Test (CBNAAT) is an automated rapid molecular diagnostic test which detects targeted Deoxyribonucleic Acids (DNA) sequences of Mycobacterium tuberculosis (M.tb) genome by Real-time Polymerase Chain Reaction (RT-PCR) method.
CBNAAT uses the GeneXpert IV Dx system and the single-use disposable Xpert M.tb/ Rifampicin (RIF) cartridges that hold the Polymerase Chain Reaction (PCR) reagents and host the PCR process.
The various TB tests utilizing the CBNAAT platform:
1) GeneXpert MTB/ RIF
- The Xpert M.tb/ RIF assay is a Nucleic Acid Amplification Test (NAAT) that uses a disposable cartridge with the GeneXpert Instrument System to quickly identify possible Multidrug-resistant TB (MDR-TB) that is resistant to RIF.
- This test yields rapid results, i.e., within 2 hours and is therefore cost and time-saving.
2) GeneXpert MTB/ RIF Ultra
- The Xpert M.tb/ RIF Ultra assay is similar to the Xpert MTB/ RIF assay, just that it is even faster with a higher level of accuracy.
- The results are obtained in 80 minutes.
- It can detect paucibacillary TB disease.
3) GeneXpert MTB/ XDR
- This is a GeneXpert M.tb assay that detects mutations associated with Resistance Towards Isoniazid (INH), Fluoroquinolones (FLQ), Second Line Injectable Drug (SLID) (amikacin, kanamycin, capreomycin) and Ethionamide (ETH) in a single test.
4) GeneXpert Omni/Edge
- GeneXpert Omni/Edge is a battery-operated, wireless and web-enabled portable molecular diagnostics system, intended for use as the point-of-care diagnostic tool in remote and challenging settings.
- It is a single-slot platform i.e., runs one test per cycle, uses a 2-in-1 tablet/laptop and a compact printer and is expected to enable accurate, fast and cost effective test results.
Apart from TB, the CBNAAT platform can be used to diagnose several other pathogens using different cartridges. One of the common other pathogens detected using CBNAAT in India is COVID-19.
Note: Except GeneXpert MTB/ RIF , other CBNAAT tests are not currently being endorsed by NTEP.
Resources
- WHO Consolidated Guidelines on Tuberculosis. Module 3: Diagnosis - Rapid Diagnostics for Tuberculosis Detection 2021 Update.
- Guidelines for Programmatic Management of Drug-resistant TB (PMDT) in India; NTEP, CTD, MoHFW, India, 2021.
- Sachdeva K, Shrivastava T. CBNAAT: A Boon for Early Diagnosis of Tuberculosis-Head and Neck; Indian J Otolaryngol Head Neck Surg, 2018.
Assessment
Question Answer 1 Answer 2 Answer 3 Answer 4 Correct answer Correct explanation Page id Part of Pre-test Part of Post-test ‘Xpert M.tb/ RIF Ultra yields results faster than Xpert M.tb/ RIF’.
True
False 1 Xpert M.tb/ RIF Ultra yields results in approx. 80 minutes compared to Xpert M.tb/ RIF which gives results in approx. 110 minutes.
Yes Yes GeneXpert MTB/ RIF is the only CBNAAT tests that is endorsed by NTEP.
True False 1 Other tests using CBNAAT platform GeneXpert MTB/ RIF Ultra, GeneXpert MTB/ XDR and GeneXpert Omni/Edge are not endorsed by NTEP. Yes
Yes
Inbuilt Controls of CBNAAT Technology
ContentThe CBNAAT System automatically performs internal quality control for each sample.
During each test, the system uses the following inbuilt controls:
System Check Control (SCC)
- Checks integrity of the instrument, cartridges and PCR reagents.
Sample Processing Control (SPC)
- Ensures that a sample is correctly processed.
- Included in the cartridge and is processed with the sample. The DNA is detected by a PCR assay.
Probe Check Control (PCC)
- Performed during the first stage of the test.
- Verifies the presence and integrity of the labelled probes.
Video fileVideo 1: CBNAAT Technology -Inbuilt Controls
Video fileVideo 2: Summary of all In-built Control Checks
Resources
Fullscreen