Preparation of Patient-wise Boxes [PWB] for DR-TB Patients
ContentUnder the National TB Elimination Program (NTEP), Drug-resistant TB (DR-TB) patients receive standardized DR-TB treatment boxes (called Patient-wise Boxes - PWB) for each month. These boxes are either with the health facility nearest to DR-TB patients (DOT centre) or with the treatment supporter who is involved in the care of DR-TB patients.
Steps involved in the preparation of second-line drug boxes for DR-TB patients:
- NTEP State Drug Store (SDS) receives quality-assured second-line drugs from the centre through the General Medical Stores Depot (GMSD) and/or Central Medical Services Society (CMSS).
- The loose medicines are utilized for individual patient-wise box creation at the SDS/DDS by the store staff.
- Each patient-wise second-line drug box is prepared according to the NTEP standard box preparation guidelines utilizing the logistics, labels and drugs, and the same details are entered in Ni-kshay Aushadi for maintaining accountability of all drug boxes.
- Each patient-wise box needs to clearly mention the batch number of different drugs it contains, the expiry of the drug box and individual drugs, and any specific instructions that need to be followed, e.g., temperature, humidity or local storage.
- Second-line drug regimen box for INH mono/ poly regimen can be prepared at the district level considering its 6-month duration with the selected 4 drugs while Bdq/ Dlm regimen-based boxes are usually prepared at SDS and provided to District Drug Stores (DDS).
The exercise of preparing the standard patient-wise boxes is done at the NTEP DDS under the guidance and supervision of the District TB Officer (DTO). In such cases, drugs to the district will be supplied in loose form through Nikshay Aushadhi.
Resources
Constituents of Monthly Patient-wise Boxes [PWB] for Shorter Oral Bedaquiline-containing MDR/RR-TB Regimen
ContentThe patient on shorter oral Bedaquiline-containing multi-drug resistant (MDR)/rifampicin resistant TB (RR-TB) regimen shall be put on Type A and Type B box when initiated on treatment on a monthly patient wise box (PWB). Bedaquiline (Bdq) needs to be issued separately and stopped after 6 months. The patient should be provided with only Type A boxes when started on continuation phase (CP).
Table 1: Constituents of monthly-type A and B PWB of shorter oral Bedaquiline-containing MDR/RR-TB regimen; Source : Guidelines for PMDT, India 2021, p.132-133 TYPE A BOX
(USE IN IP AS WELL AS CP)
Drugs
Strength
16-29 kg
30-45 kg
46-70 kg
>70 kg
Tab. Levofloxacin#
250/500 mg
250 mg *30tab
250 mg *30tab + 500 mg *30tab
500 mg *60tab
500 mg *60tab
Tab. Clofazimine
50/100 mg
50 mg *30tab
100 mg *30tab
100mg *30tab
100 mg *60tab
Tab. Ethambutol
400/800 mg
400 mg *30tab
800 mg *30tab
400 mg *30tab + 800 mg *30tab
800 mg *60tab
Tab. Pyrazinamide
500/750 mg
750 mg *30tab
500 mg *30tab + 750 mg *30tab
500 mg *60tab + 750 mg *30tab
500 mg *30tab + 750 mg *60tab
Tab. Pyridoxine
50/100 mg
50 mg*30tab
100 mg*30tab
100 mg*30tab
100 mg*30tab
Type B Box
(Use in IP)
Drugs
Strength
16-29 kg
30-45 kg
46-70 kg
>70 kg
Tab. Isoniazid
100/300 mg
300 mg *30tab
300 mg *60tab
300 mg *90tab
300 mg *90tab
Tab. Ethionamide
125/250 mg
125 mg *30tab + 250 mg *30tab
250 mg *60tab
250 mg *90tab
250 mg *120tab
Bedaquiline bottle
100 mg
1 Jar (Jar of 188 tablets for full course)
# When moxifloxacin prescribed under exceptional condition instead of levofloxacin, the modified box with moxifloxacin (normal dose) can be prepared from standard box at district drug store (DDS)
Resources
Kindly provide your valuable feedback on the page to the link provided HERE
Constituents of Patient-wise Boxes [PWB] for Isoniazid [H] Mono/Poly DR-TB Regimen
ContentStandard Patient-wise Boxes (PWBs) are constituted for TB patients initiated on Isoniazid (H) Mono/ Poly Drug-resistant TB (DR-TB) regimen.
This regimen has no segregation in terms of Intensive Phase (IP) or Continuation Phase (CP), hence drugs are provided in a single type of PWB.
Table: Constituents of standard PWB (6 months) of H mono/ poly DR-TB regimen; Source: Guidelines for PMDT, India 2021, p134
STANDARD PWB
CONTINUE FOR COMPLETE TREATMENT Drugs
Strength
16-29 kg
30-45 kg
46-70 kg
>70 kg
Tab. Levofloxacin
250/ 500 mg
250 mg *180 tab
250 mg *180 tab + 500 mg *180 tab
500 mg *360 tab
500 mg *360 tab
Tab. Rifampicin
150/ 300/ 450 mg
300 mg *180 tab
450 mg *180 tab
300 mg *360 tab
300 mg *360 tab + 150 mg *180 tab
Tab. Ethambutol
400/ 800 mg
400 mg *180 tab
800 mg *180 tab
400 mg *180 tab + 800 mg *180 tab
800 mg *360 tab
Tab. Pyrazinamide
500/ 750 mg
750 mg *180 tab
750 mg *180 tab + 500 mg *180 tab
750 mg *180 tab + 500 mg *360 tab
750 mg *360 tab + 500 mg *180 tab
Tab. Pyridoxine
50/100 mg
50 mg *30 tab
100 mg *30 tab
100 mg *30 tab
100 mg *30 tab
*No separate box for IP and CP.
Resources
- Guidelines for Programmatic Management of Drug-resistant Tuberculosis in India, March 2021.
- Central TB Division, MoHFW. Training Modules (5-9) for Programme Managers & Medical Officers, 2020.
- Standard Operating Procedure Manual - Procurement & Supply Chain Management, RNTCP.
- Technical and Operational guidelines for TB in India, 2016.
Kindly provide your valuable feedback on the page to the link provided HERE
Management of Patient-wise Boxes in Different Scenarios
ContentThere are different scenarios regarding patient-wise box (PWB) management. These are described below.
Scenario 1: Modification in regimen
In this scenario, the nodal and district drug-resistant tuberculosis centre (N/DDR-TBC) committee decides on a regimen modification.
- The district drug store (DDS) prepares modified PWBs from standard available boxes and loose drugs and arranges the supply of these PWBs to the treatment supporter (TS).
- The district TB officer (DTO) ensures that the drugs are supplied as per the modified regimen for all subsequent months.
Scenario 2: Extension of intensive phase
If intensive phase (IP) is extended:
- N/DDR-TBC informs the DTO
- DTO informs the health facility (HF) doctor and respective TB unit (TU)
- HF releases 1-month PWB to the respective treatment support centre from where the patient is taking treatment
When the patient is switched to continuation phase (CP) in case of a shorter regimen:
- DTO, again, informs HF doctor and respective TU
- HF releases 1-month PWB to the respective treatment support centre from where the patient is taking treatment
In the case of a longer regimen, after completion of 6-8 months of treatment, Linezolid (Lzd) should be reduced to 300 mg following N/DDR-TBC directives.
All patients must complete their monthly boxes before switching to the subsequent box provided.
Scenario 3: Change in DR-TB regimen
If N/DDR-TBC decides to change the DR-TB regimen of a patient, the DDS supplies a new treatment regimen box from PWB/ loose drugs. Unused drugs including Bdq containers should be sent back to the DDS. In this scenario, the patient should be immediately switched to the new regimen designed by N/DDR-TBC.
Resources
Kindly provide your valuable feedback on the page to the link provided HERE
Role of the State Drug Store [SDS] in the Constitution of Patient-wise Boxes
ContentThe State Drug Store (SDS) shall constitute drug boxes for:
- Shorter oral Bedaquiline (Bdq)-containing Multi-drug resistant (MDR)/ Rifampicin-resistant Tuberculosis (RR-TB) regimen (Type A and B)
- Longer oral Multi (M)/ Extensively Drug-resistant Tuberculosis (XDR-TB) regimen
- Isoniazid (H) mono/ poly DR-TB regimen
Boxes will be prepared manually only and not through Nikshay Aushadhi software.
From Nikshay Aushadhi drugs will be supplied in loose to districts.
Based on the requirement of districts SDS will supply boxes to respective districts by a hired transport agency or other mechanisms.
Loose drugs will also be supplied from SDS to the District Drug Store (DDS) for modification and preparation of new boxes.
Resources
Kindly provide your valuable feedback on the page to the link provided HERE
Role of the District Drug Store [DDS] in the Constitution of Patient-wise Boxes
ContentWhen there is a modification in the Drug-resistant TB (DR-TB) regimen as suggested by the Drug-Resistant Tuberculosis Centre (DR-TBC), the district drug storekeeper/ pharmacist prepares the modified boxes from loose Second-line anti-TB Drugs (SLD) supplied from the State Drug Store (SDS).
- The state shall provide the necessary support for capacity building of the District Drug Store (DDS) for carrying out the entire exercise of preparing standardized/ modified patient-wise drug boxes at the DDS level.
- Whenever oral regimens are modified during the course of treatment, the DDS needs to ensure that the change in the regimen should be incorporated into the supply of subsequent boxes.
- A full-time DDS storekeeper/ pharmacist must be mandatorily recruited/ placed for a successful decentralized system of preparation of drug boxes at the DDS level.
- District TB Officer (DTO) and Medical officer-DTC usually perform a random check to understand if there is any challenge in the constitution of patient-wise boxes.
Resources
Kindly provide your valuable feedback on the page to the link provided HERE
Patient-wise Drug Boxes: Packing Instructions
ContentPackaging of loose drugs into monthly patient-wise boxes should be done under the guidance of those in charge of drug logistics at the state and district levels.
Patient-wise drug box preparation should be done for the following Drug-resistant TB (DR-TB) regimens:
- Shorter oral Bedaquiline-containing Multidrug-resistant/ Rifampicin-resistant TB (MDR/RR-TB) regimen
- Longer oral Multi (M)/ Extensively Drug-resistant TB (XDR-TB) regimen
- Isoniazid (H) mono/poly DR-TB regimen
Packaging Instructions
Patient-wise drug boxes are prepared as per the type of DR-TB regimen and weight bands. One monthly pouch of the capsule Cycloserine (Cs) and tablet Ethambutol (E) each, should be made from a plastic bag with a zip lock facility in which 1 gm pouch of silica gel desiccant should be kept.

Figure 1: Isoniazid (H) Mono/ Poly Regimen DR-TB Treatment Box
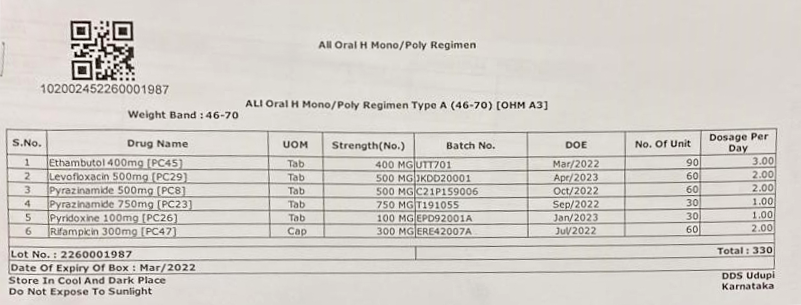
Figure 2: Isoniazid (H) Mono/ Poly Regimen DR-TB Label
In each Type A box, one pouch of silica gel desiccant of 4 gm weight should also be kept.
Labels for the boxes should be developed with the following information (Figures 2 and 3):
- Item-wise name of drugs with a quantity of each drug in the box
- Batch number and Date of Expiry (DOE) of the individual drugs
- DOE of boxes, with expiry date of the drug having the shortest expiry date
- Date of issue of the box from the State Drug Store (SDS)
- Serial number of the box
- Storage instructions on the box for ensuring adequate precautions in storage of the drugs, especially at the treatment supporter level. Some suggested messages are:
- “Store in a cool and dark place, preferably in a clean cupboard”
- “Do not expose to direct sunlight”
- “Keep away from children/unauthorized persons”
- “Box to be closed properly every time after the withdrawal of drugs”.
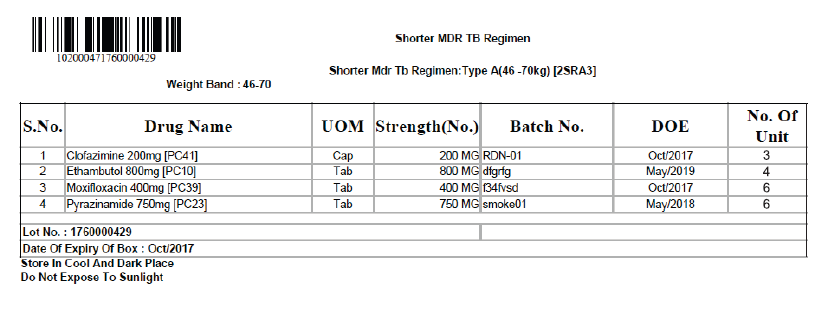
Figure 3: Prototype of a Label (Type A box for shorter DR-TB regimen for the weight band - 46 to 70 kg)
Resources
- Guidelines for Programmatic Management of Drug-resistant Tuberculosis in India, March 2021.
- Standard Operating Procedure Manual - Procurement & Supply Chain Management, MOHFW, GOI.
Kindly provide your valuable feedback on the page to the link provided HERE
Fullscreen