-
CDST_LT: Basics of microscopy
FullscreenComparing AFB and LED-Fluorescence Microscopy
ContentThe comparison of the Acid-Fast Bacilli (AFB) and Light Emission Diode (LED) Fluorescence Microscopy is tabulated below:
Direct Microscopy for Acid Fast Bacilli (AFB)
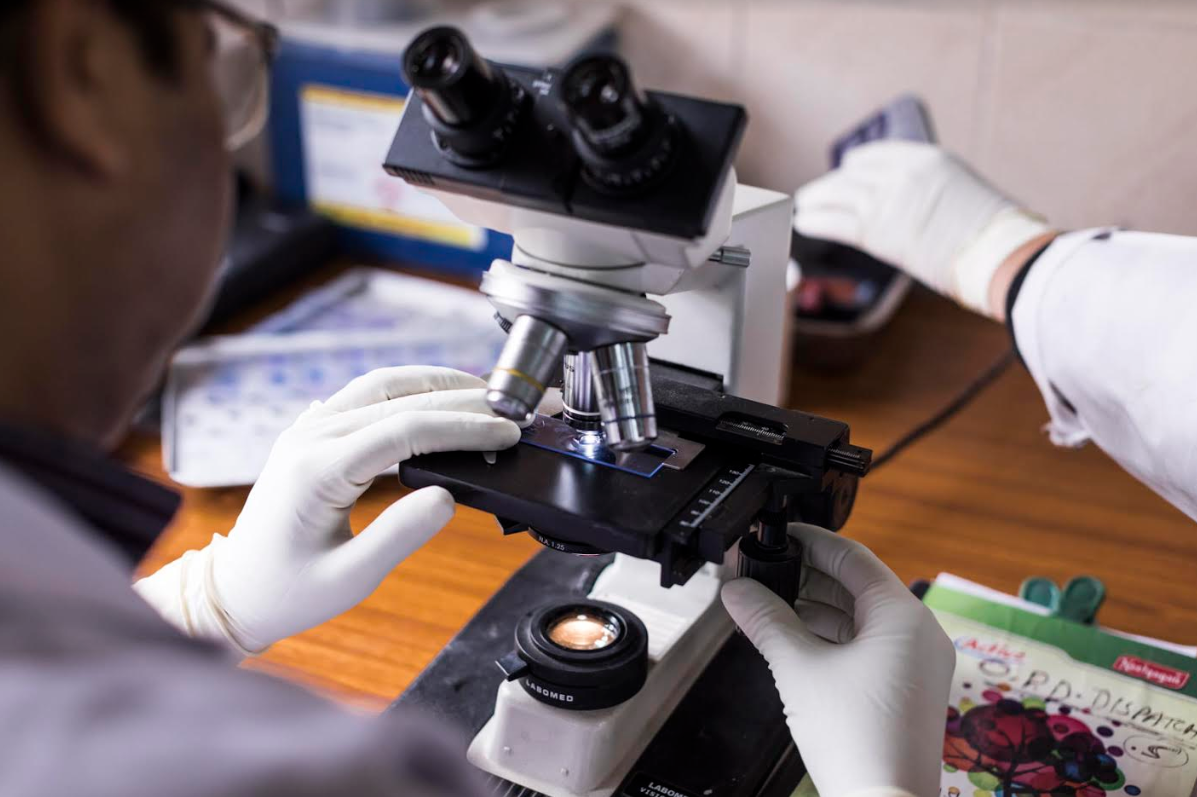
Figure 1: Bright Field Microscope used in Ziehl–Neelsen (ZN) staining of AFB
Light Emission Diode (LED)
Fluorescence Microscopy
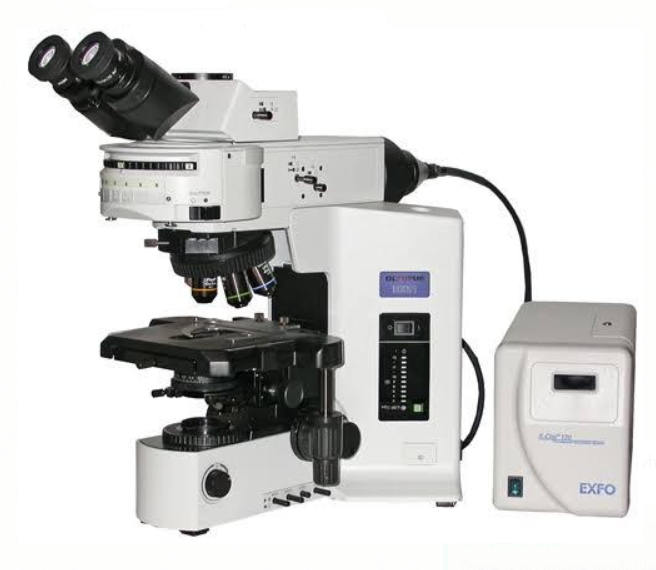
Figure 2: LED-Fluorescent Microscope used in fluorescent staining of AFB
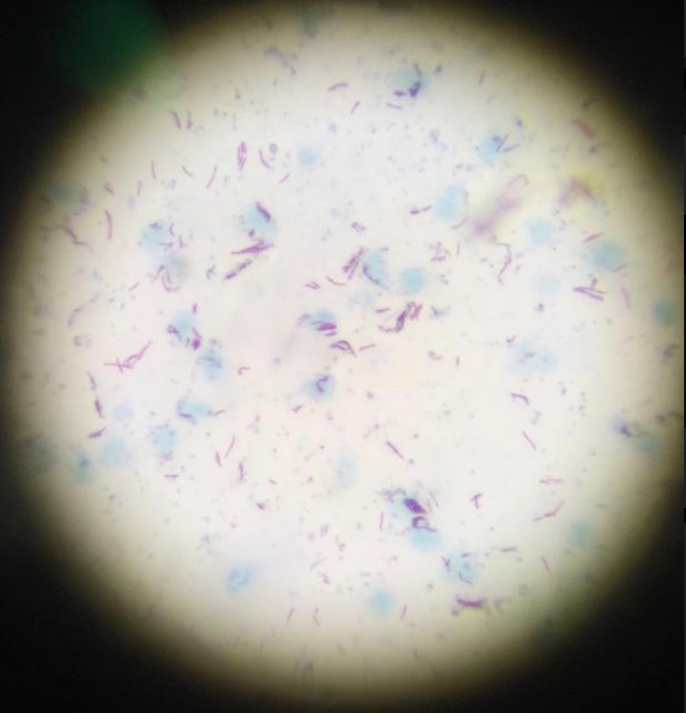
Figure 3: ZN-stained slide used in direct microscopy


Figure 4: Auramine-O-stained slide used in fluorescent microscopy
Method used widely for the diagnosis and confirmation of pulmonary tuberculosis
Method used less commonly for the diagnosis and confirmation of pulmonary tuberculosis
Staining procedure complex
Staining procedure simpler
Examination at lower magnification is NOT possible
Examination at lower magnification is possible
Takes more time than LED-FM
Takes 75% less time than ZN and chances to lose scanty slides are also minimized.
Low durability
High durability
Less sensitive
10% more sensitivity than Bright Field ZN microscopy
As specific as LED-FM
Equally as specific as ZN microscopy
No need for an experienced lab technician
Needs an experienced lab technician
Technical errors are common
Technical errors are less common
Misses the paucibacillary TB cases, especially when the patient is co-infected with HIV
Identifies the paucibacillary TB cases especially when the patient is co-infected with HIV
Resources
- WHO Policy Statement - Fluorescence Light Emitted Microscope for the Diagnosis of TB, 2010.
- A Comparative Study of Auramine Staining using LED Fluorescent Microscopy with Ziehl- Neelsen Staining in the Diagnosis of Pulmonary Tuberculosis, Journal of Evolution of Medical and Dental Sciences, Volume 2, Issue 20, May 2013.
- Comparison of Direct versus Concentrated Smear Microscopy in Detection of Pulmonary Tuberculosis, BMC Res Notes, 2013; 6 : 291.
Kindly provide your valuable feedback on the page to the link provided HERE
Ideal Infrastructure Required for Microscopy Centers
ContentUnder the National Tuberculosis Elimination Program (NTEP) network, the most peripheral laboratories are the Designated Microscopy Centres (DMC) which serve a population of around 100,000 each (50,000 population norm in tribal and hilly areas).
The basic requirements for microscopy centres include (Figure):
- Good ventilation by exhaust fan and open windows
- A table/ work bench to prepare smears
- A sink to stain smears
- A table/ bench to examine smears
- A table/ bench for documentation
- Basin for hand washing with supply of water
- Good lighting
- Non-slip flooring
- Sample receiving area
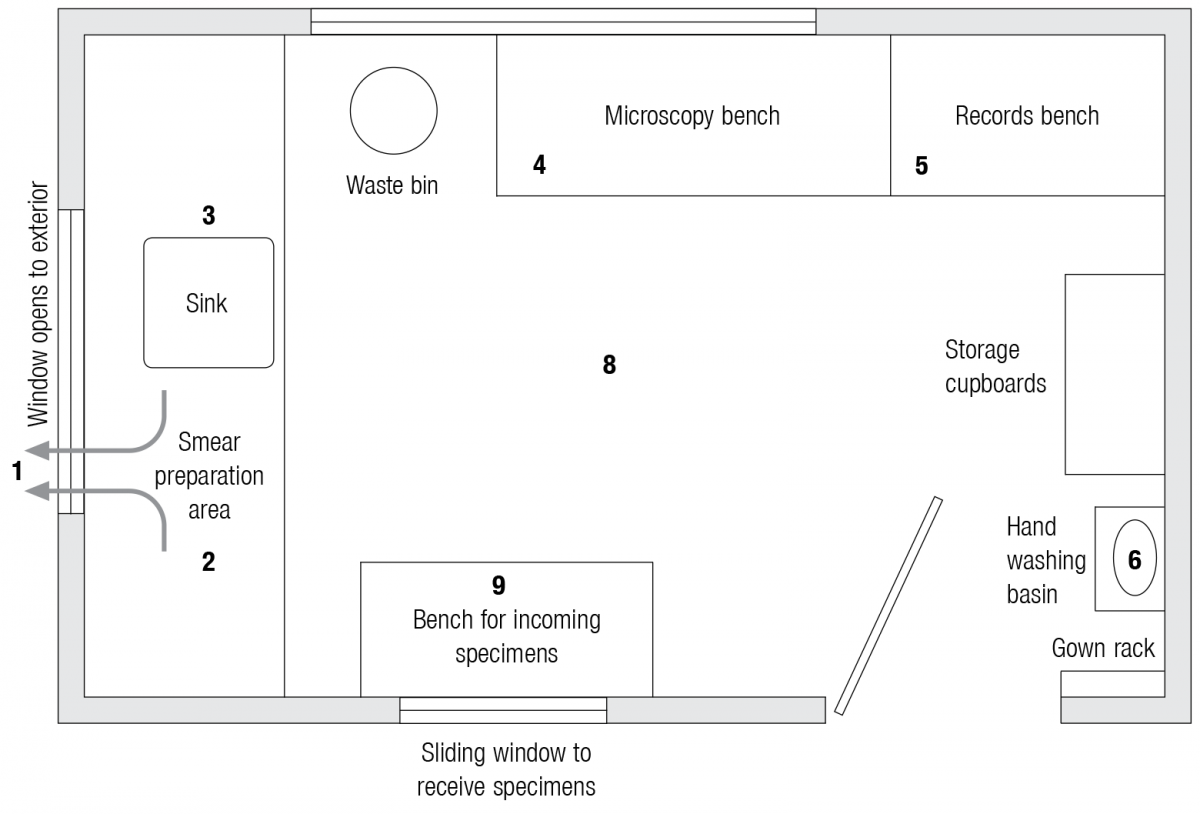
Figure: Ideal Infrastructure required for Microscopy Centres
Laboratory Diagnosis of Tuberculosis by Sputum Microscopy
ESTABLISHMENT
PROVISIONS UNDER NTEP FOR DMC FACILITIES
Infrastructure development
Normative amount as per “Norms and basis of costing” for NTEP. Unit cost for initial establishment/ refurbishment/ upgradation/ maintenance of civil work to be carried out as per rates prescribed by Public Works Department (PWD) or cell/ division/ corporation/ wing for infrastructure development
Human resources
NTEP trained Lab Technician (LT)
Diagnosis
- Provision for sputum collection in the presence or absence of an LT
- Adequate space for storing and using binocular microscope, essential lab consumables, space for recording and reporting
- Availability of running water
- Availability of electricity
- Availability of biomedical waste management facilities
Equipment
Binocular microscope with a light source
Location of DMC
Ideally, the DMC should be located in a peripheral health facility that has at least 50 new adult OPD/day.
Resources
- Technical and Operational Guidelines for TB Control in India, 2016.
- Laboratory Diagnosis of Tuberculosis by Sputum Microscopy, GLI Initiative.
Kindly provide your valuable feedback on the page to the link provided HERE
Microscopy
ContentMicroscopy is a TB diagnostic technology that utilizes the acid-fastness property of Mycobacterium tuberculosis to visualize it under a microscope. Results of sputum smear microscopy can either be smear-negative, or smear-positive (with various grades).
Advantages:
- It is currently the most accessible and cheapest TB diagnostic test available under National TB Elimination Programme (NTEP) in India.
- It has the shortest turnaround time for diagnosis.
- It has high specificity.
Limitations:
- Low sensitivity. It becomes positive only when more than 5000 bacilli/ml of sample are present. Hence, cases would be missed in early disease, or when an inappropriate biological specimen is provided, where bacterial load in sputum is less.
- It is unable to differentiate between M. tuberculosis and Non-tuberculous Mycobacteria (NTM). This is predominantly an issue in geographies with lower burden.
There are two types of microscopies used in NTEP: Ziehl-Neelsen (ZN) Microscopy and Fluorescence Microscopy (FM). These vary in the type of stain and microscope used. FM is newer of the two types and is currently recommended for use over ZN.
Resources
Parts of a Microscope
ContentVideo fileWorking with a Microscope
ContentVideo file
-
CDST_LT: Sputum smear preparation
FullscreenSteps involved in Smear Microscopy
ContentSputum smears must be prepared promptly after samples are collected or received in the laboratory.
Steps in smear preparation are as follows:
- Cleaning and Labelling of slide (No 1)
- Making the smear (No 2-3)
- Drying and Heat fixing the smear (No 4-5)
- Staining and counterstaining the smear (No 6- 12)
- Examination of slide/Reading the Smear (No 13-14)
- Reporting and recording the observations (Digitally and TB Lab register)
- Storage of slides (as per Laboratory Numbers in closed box) (No 15)


Figure: Steps in Smear Preparation; 1- Labelling of the slide, 2- Using a broomstick to pick up purulent portion (A) while avoiding the salivary portion (B), 3- Spreading sample on a glass slide, 4- Air-drying the slides, 5- Heat-fixing the smeared slides,6- Staining with 1% Carbol fuchsin, 7- Heating of stained smear, 8- Decolorize with 25% Sulphuric acid, 9- Rinse off decolourizer, 10- Counter stain with 0.1% Methylene blue, 11-Rinse off counter stain, 12-Drying the prepared slide, 13-14 Examination of smears , reporting of observations, 15-Storage of slides; Source: Laboratory Diagnosis by Sputum Smear Microscopy.
Resources
- Laboratory Diagnosis by Sputum Smear Microscopy - The Handbook, GLI, 2013.
- Module for Laboratory Technicians, CTD, 2005.
Question Answer 1 Answer 2 Answer 3 Answer 4 Correct answer Correct explanation Page id Part of Pre-test Part of Post-test Sputum smears should be dried by heating. True False 2 Sputum smears should be air-dried. Yes Yes Clean, fresh, unscratched slides should be used for smear preparation.
True False 1 Prevents deposits on stained smear. Yes Yes Cleaning and Labelling Slide
ContentA Laboratory Serial Number is assigned to each presumptive TB patient who is examined at the microscopy centre.
Each Laboratory Technician (LT) needs to ensure that all the slides are labelled using the Laboratory Serial Number. This is essential for recording as well as for the review of the slide during the supervisory visit as well as during the quality assurance exercise.
For every test, a new slide needs to be used. It is essential that there are no fingerprints or any scratches on the side of the slide (see figure 1).

Figure 1: Always select new, clean, grease-free and unscratched slides
Once the LT is ready to prepare a smear, he/she needs to write the Laboratory Serial Number on the left side of the slide with a diamond marker or a grease pencil only (see figure 2). Avoid multiple labelling (see figure 3).
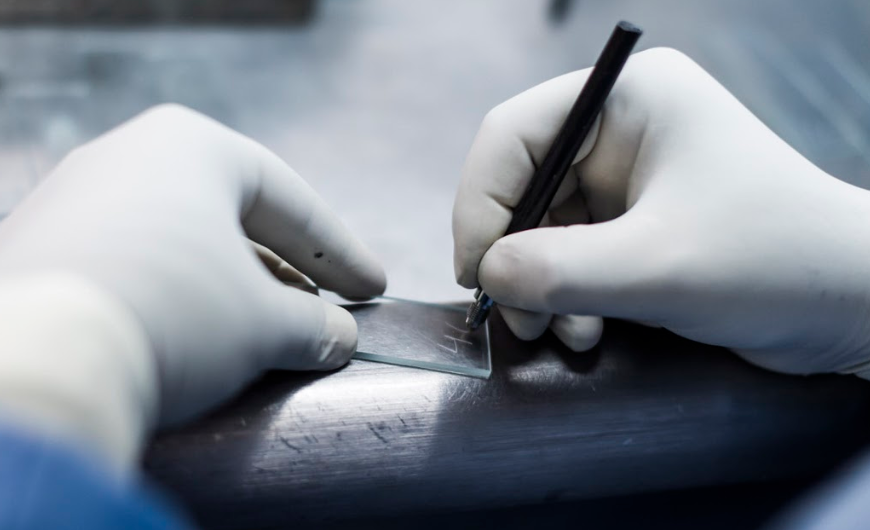
Figure 2: The laboratory serial number is written on one end of the slide using a diamond marker

Figure 3: Avoid Multiple Labelling; a grease pencil has been used to label the slide.
Labelling of slides needs to be monitored and supported by the concerned Senior TB Laboratory Supervisor (STLS) during External Quality Assessment (EQA) visits.
Resources
Kindly provide your valuable feedback on the page to the link provided HERE
Making the smear
ContentThe National TB Elimination Programme (NTEP) has a standard procedure for sputum smear preparation, the steps for which are listed below:
The tools required for smear preparation include a clean work surface, new and clean glass slides, a discard bucket or a foot-operated bin with a plastic liner, bamboo or wooden applicator sticks or sterile wire loop, spirit lamp and a rack for drying smears.
- Use new, clean, unscratched glass slides and label the slide with the laboratory serial number.
- Prepare the smear in the centre of the slide covering 3 cm X 2 cm
The smear is prepared by using either a wooden stick (Figure 2) or a sterile wire loop (Figure 3).

Figure 2: Wooden stick used in smear preparation

Figure 3: Sterile wire loop used in smear preparation
Steps for Smear Preparation Using a Wooden Stick
- Break the wooden stick into two halves with uneven ends.
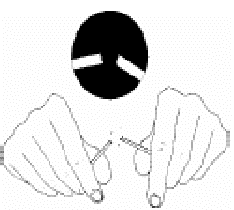
- Using the uneven end, select and pick purulent portions of the sputum specimen and transfer onto a new, clean, labelled, glass slide.
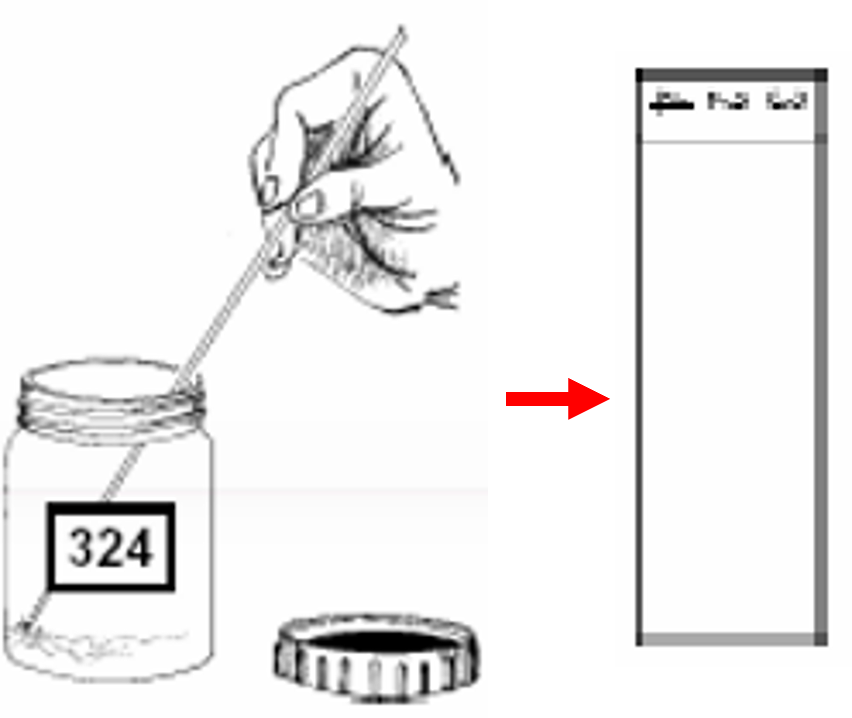
- Using the wooden stick, spread the sputum evenly, in a continuous rotational movement, to cover two-thirds of the central portion of the slide. Smear preparation should be done near a flame. This is required as approximately 6 inches around the flame is considered as a sterile zone which coagulates the aerosols raised during smear preparation.
- Discard the used wooden sticks in the discard bucket or a foot-operated bin with a liner and disinfectant. A different broomstick is used for each smear so that one patient's sputum is not mixed with another patient's sputum.

- Air-dry the smear slide on the rack for 30 minutes
- After air-drying, heat-fix the smear, using a lighted spirit lamp.
Steps for Smear Preparation Using a Wire Loop
- Take a nichrome wire loop or a disposable loop.

- Using the loop, select and pick purulent portions of the sputum specimen and transfer onto a new, clean, labelled, glass slide.
- Using the loop, spread the sputum evenly, in a continuous rotational movement, to cover two-thirds of the central portion of the slide.

- After use, sterilize the loop in an electric loop sterilizer or flame the loop to red-hot.


- Air-dry the smear slide on the rack for 30 minutes.
- After air-drying, heat-fix the smear using a lighted spirit lamp.
Resources
- Module for Laboratory Technicians (RNTCP), Central TB Division, MoHFW, 2005.
- Laboratory Diagnosis of Tuberculosis by Sputum Microscopy, GLI Initiative.
Kindly provide your valuable feedback on the page to the link provided HERE
Air drying and heat fixing
ContentFor sputum smear microscopy, the slides should be air-dried as heating the slide while the smear is wet can result in bubbling of TB bacilli into the air.
Fixation makes the sputum stick to glass slide and preserves the shape of the bacilli.
The procedure for air-drying and heat-fixing the slide is as follows:
- A smear prepared on a clean glass slide from mucopurulent portion of the specimen is air dried for 15-30 minutes on a rack (see Figure 1)

Figure 1: Rack for air-drying slides
- When dry, the smear facing upwards is fixed by heat from below. This can be achieved by passing the slide 2-3 times over the flame of a spirit lamp (as shown in Figures 2 and 3) for 3-4 seconds each time.

Figure 2: Fixing the Smear by Heat Fixation; Source: Laboratory Diagnosis by Sputum Smear Microscopy

Figure 3: Spirit lamp used to heat-fix smears
Important points to consider when fixing smears
- Heat fixing does not always kill Mycobacteria, exercise care when handling slides.
- Flame fixing may aerosolize bacilli from the smear.
- Overheating can damage the bacilli, burn the smear or break the slide.
- Insufficient heat or time can lead to smear washing off during staining steps.
- Heating for too short a period can result in a false-negative result because the TB bacilli will not be well preserved on the slide.
After the smears are fixed, they can be stained for examination or stored, or used in proficiency testing panel and quality control slides for staining.
Resources
Laboratory diagnosis by sputum smear microscopy
Assessment
Question
Answer 1
Answer 2
Answer 3
Answer 4
Correct answer
Correct explanation
Page id
Part of Pre-test
Part of Post-test
Heat fixing does not always kill mycobacteria.
True
False
1
Heat fixing does not always kill mycobacteria.
Yes
Yes
Qualities of a good sputum smear
ContentGood quality smears are essential for accurate examination and results. A good quality sputum smear is one that is of uniform thickness and made from the mucopurulent portion of the sample in the center of the slide (Figure 1).
Do’s and Don’ts of a Good Quality Smear
- Do ensure that the smear size is 3 cm by 2 cm
- Do ensure there are no fingerprints on the prepared smear

Figure 1: Uniform spread of smear, not too thick or thin, and covering an area of 3cm by 2cm
- The smear should not be very thick, but it should be thin enough to visualize a newsprint as can be seen in Figure 2

Figure 2: Smear thin enough to visualise a print through it
- All smears should be air-dried for 30 minutes, before heat fixing to ensure that the smear is not washed off during staining
Common Mistakes to Be Avoided

Resources
- The Handbook - Laboratory Diagnosis of Tuberculosis by Sputum Microscopy, 2013
- Module for Laboratory Technicians – RNTCP, 2005
- Manual for Sputum Smear Fluorescence Microscopy- RNTCP
Kindly provide your valuable feedback on the page to the link provided HERE
-
CDST_LT: Staining sputum smears
FullscreenZN Microscopy: Staining Process
ContentAfter air drying and heat-fixing of the smear, it needs to be stained for the identification of MTB during microscopy.
The reagents required for the ZN staining procedure are shown in Figure 1. The steps involved in the staining procedure are shown in Figure 2.

Figure 1: Reagents Required for ZN Staining

Figure 2: Steps involved in ZN staining procedure
For a visual representation of the above-mentioned steps, please click the video below.
Video fileResources
Laboratory Diagnosis by Sputum Smear Microscopy, GLI Initiative
Assessment
Question Answer 1 Answer 2 Answer 3 Answer 4 Correct answer Correct explanation Page id Part of Pre-test Part of Post-test When staining ZN slides, it is important to heat the slides until it begins to boil. True False 2 When staining ZN slides, it is important to heat the slides gently until you see vapours, but do not boil. Yes Yes ZN Microscopy: Properties of a well stained slide
ContentCharacteristics of a Well-stained Slide
- Staining and appearance of Mycobacterium species in ZN staining.
- The primary dye - carbol-fuschin stains all cells pink in sputum samples.
- The bacilli retain the primary pink carbol-fuschin on decolourisation with acid-alcohol.
- Epithelial, pus and mucous cells in the sputum decolourise on the addition of the acid-alcohol and take up the counterstain dye of methylene blue.
- Thus, Mycobacterium species appears pink against a background of epithelial, pus and mucous cells that appear blue.
- ZN-stained Mycobacterium species are visible as pink, long, slender rods with granules or V-shaped or in clumps.
- Mycobacterium species should not be stained too dark or pale pink.
- Possible reasons:
- Smear thickness is not appropriate
- Insufficient decolourisation
- Low acid concentration
- Carbol-fuchsin dries on smear
- Primary staining/ decolourisation time not appropriate
- Expired reagents used
- Possible solutions:
- Internal quality control of prepared smears and stains
- Use a stopwatch to time staining steps.
- Do not overheat carol-fuchsin.
- Possible reasons:
- The counter-stained cells should not be dark blue.
- Possible reasons:
- Smear thickness is not appropriate
- Excessive counterstaining time
- Inadequate washing after counterstaining
- Methylene blue concentration is not appropriate
- Possible solutions:
- Internal quality control of prepared smears and stains
- Use a stopwatch to time staining steps
- Possible reasons:
- There should not be any deposits on stained smears.
- Possible reasons:
- Stains not filtered
- Slides not clean
- Possible solutions:
- Internal quality control of prepared stains
- Filter stains
- Use clean, fresh, unscratched slides for smear preparation.
- Possible reasons:
Resources
- Laboratory Diagnosis of Tuberculosis by Sputum Smear Microscopy - The Handbook, GLI, 2013.
- Module for Laboratory Technicians, RNTCP, CTD, 2005.
Assessment
Question Answer 1 Answer 2 Answer 3 Answer 4 Correct answer Correct explanation Page id Part of Pre-test Part of Post-test Which of these is/are the characteristic/s of a well-stained slide? Mycobacterium species should not be stained too dark or pale pink. Mycobacterium species are visible as pink long slender rods with granules or V-shaped or in clumps. The counter-stained cells should not be dark blue. All the above 4 ZN-stained Mycobacterium species are visible as pink, long, slender rods with granules or V-shaped or in clumps. The bacilli should not be stained too dark or pale pink. The counter-stained cells should not be dark blue. Yes Yes - Staining and appearance of Mycobacterium species in ZN staining.
Fluorescent Microscopy: Staining Process
ContentAfter air drying and heat-fixing of the smear, it needs to be stained.
Fluorescence microscopy (FM) staining should always be carried out in a designated area. The reagents required for the FM staining procedure are shown in Figure 1. The steps involved in the staining procedure are shown in Figure 2.

Figure 1: Reagents Required for FM Staining


Figure 2: Steps involved in FM staining procedure
For a visual representation of the above-mentioned steps, please click the video below:
Video fileResources
Laboratory Diagnosis by Sputum Smear Microscopy, GLI Initiative
Assessment
Question
Answer 1
Answer 2
Answer 3
Answer 4
Correct answer
Correct explanation
Page id
Part of Pre-test
Part of Post-test
What is the counterstain used in FM staining?
Acid Alcohol
Potassium Permanganate
Phenol
Auramine-O
2
In FM staining, Potassium Permanganate is used as the counterstain.
Yes
Yes
Fluorescent Microscopy: Properties of a well stained slide
ContentLight-emitting Diode Fluorescence Microscopy (LED-FM) utilises the fluorescent dye Auramine-O to stain and detect Mycobacterium species in clinical samples.
Characteristics of a Well-stained Slide
- Auramine-O stained Mycobacterium species are visible as bright yellow/ green long slender rods, slightly curved, with variable lengths, single or in clumps, with uniform staining or granular appearance against a dark background (Figure A).
-
Slides stained with Auramine-O should not have too much background fluorescence (Figure B)
Possible reasons:
- Smear thickness is not appropriate
- Insufficient decolourisation
- Counterstain too weak
- Auramine-O dries on the smear
-
Auramine-O not filtered
Possible solutions:
- Internal quality control of prepared smears and stains
- Use a stopwatch to time-staining steps
- Filter Auramine-O before use
- Add a sufficient quantity of stains to cover the smear
-
Slides stained with Auramine-O should not have pale fluorescence (Figure C)
Possible reasons:
- Smear thickness is not appropriate
- Low Auramine-O concentration
- Excessive decolourisation time
-
Stained smears exposed to daylight
Possible solutions:
- Internal quality control of prepared smears and stains
- Use a stopwatch to time-staining steps
- Store Auramine-O and stained slides in the dark
- Read stained slides as early as possible
- Non-fluorescent yellow/ green coloured bacillary shapes should not be considered as Mycobacterium species.
-
Slides stained with Auramine-O may contain stained artefacts/ background debris which are not Mycobacterium species.

Figure: Slides stained with Auramine-O showing bright yellow/ green slender rods (A); slide with too much background fluorescence (B); slide with pale fluorescence (C); Source: Laboratory Diagnosis by Sputum Smear Microscopy
Resources
- Laboratory Diagnosis of Tuberculosis by Sputum Smear Microscopy - The Handbook, GLI, 2013.
- Manual for Sputum Smear Fluorescence Microscopy, RNTCP, CTD.
Assessment
Question Answer 1 Answer 2 Answer 3 Answer 4 Correct answer Correct explanation Page id Part of Pre-test Part of Post-test Which of these is/are the characteristic/s of a well-stained FM slide? Mycobacterium species are visible as bright yellow/ green long slender rods against a dark background. Mycobacterium species are visible as pink, long slender rods.
Mycobacterium species are visible as pale yellow/ green long slender rods against a dark background. Mycobacterium species are visible as non-fluorescent yellow/ green coloured bacillary shapes. 1 Mycobacterium species are visible as bright yellow/ green long slender rods against a dark background. Yes Yes
-
CDST_LT: Reading smears and reporting microscopy results
FullscreenProcess of Reading a Smear
ContentThe manner and quality of smear reading has a major impact on the result of sputum smear microscopy and case detection. Each slide needs to be examined for at least 5 full minutes or 100 fields need to be examined.
The overall process of reading a smear is outlined in the figure below:

Figure: Process of reading a smear
Resources:
- AFB Smear Microscopy, Trainer Notes.
- Module for Laboratory Technicians.
- Laboratory Diagnosis of Tuberculosis by Sputum Microscopy.
Assessment
Question
Answer 1
Answer 2
Answer 3
Answer 4
Correct answer
Correct explanation
Page id
Part of Pre-test
Part of Post-test
What is the minimum time to be spent by an LT when examining a smear under the microscope?
30 minutes
5 minutes
2 minutes
30 seconds
2
Even the most experienced microscopist needs to examine each slide for at least 5 full minutes.
Oil immersion and focusing
ContentThe following are the measures to be kept in mind when adding immersion oil to the slide:
- Before adding immersion oil to the slide, ensure that the smear is facing upwards (Figure 1).

Figure 1: Slide should Face Upwards when Mounted on the Microscope Stage
- Add a drop of immersion oil, using the applicator (dropper bottle).
- The drop must fall freely onto the smear, so that the oil applicator does not touch the slide and get contaminated (Figure 2).

Figure 2: Oil Dropped onto Slide with an Applicator
- Mount the slide on the microscope and use the 10X objective lens to focus the smear, scan and look for mucoid material.
- Carefully rotate the 100X objective lens over the slide, adjust, sharp focus and observe under immersion oil.
Do’s and Don’ts:
- The 100X objective is the only lens requiring immersion oil.
- Keep the immersion oil away from other lenses.
- Use a good quality immersion oil.
- The immersion oil must have a medium viscosity and a refractive index (RI) greater than 1.5.
- Never allow the oil applicator to touch the slide.
- Do not use cedar wood oil diluted with xylene as it leaves a sticky residue on the lens and destroys the lens over time.
Resources
- Guidelines for Laboratory Consumables, Central TB Division, 2019.
- Laboratory Diagnosis of Tuberculosis by Sputum Microscopy, GLI Initiative.
Kindly provide your valuable feedback on the page to the link provided HERE
Reading a ZN stained smear
ContentImageResources
- AFB Microscopy Trainer Notes
- Module for Laboratory Technicians
- Laboratory diagnosis by sputum smear microscopy
Kindly provide your valuable feedback on the page to the link provided HERE
Reading a Fluorescent Stained slide
ContentSmears stained with the fluorescent dye, Auramine, are read with a Light Emitting Diode Fluorescence Microscope (LED-FM).
- The fluorescent-stained smears are to be read within 24 hours of staining in a dark room as the fluorescent stain fades on exposure to light.
- The slides are to be stored in the slide box to avoid exposure to light. Alternatively, they may be stored wrapped in brown or black paper and kept away from light.
- The slides should not be stored in a fridge as 4°C does not prevent or delay fading of fluorescent dye.
After fluorescent staining, smears are examined at much lower magnifications (typically 200x) than used for Ziehl Neelsen (ZN)-stained smears (1000x). Each field examined under fluorescence microscopy, therefore, has a larger area than that seen with bright field microscopy.
Examination Procedure
The steps include:
1. Switch on the LED-FM in a dark room.
2. Focus the stained slide using a 20x or 40x objective lens and view through the 10x eyepiece (magnification of 200x or 400x).
- Does not require the use of oil immersion.
3. Read smear in a linear pattern.
4. Count bacilli that appear as slender bright yellow fluorescent rods against a dark background in 30-50 fields. Rule out any artefact.
- For a trained and experienced Lab Technician (LT), each smear would take approximately 2 minutes for 30-50 fields or three horizontal sweeps.
Reporting Procedure
The smears are reported as negative/ scanty/ 1+/ 2+/ 3+ per the grading scale (Table).
Table: A Comparison between ZN (1000x) and FM (400x and 200x) for Grading of Smears at Different Magnifications
Result
1000 x magnification
1 length=100 HPF
400x magnification
1 length= 40 fields= 200 HPF
200x magnification
1 length=30 fields=300 HPF
Negative
Zero AFB/1 length
Zero AFB/1 length
Zero AFB/1 length
Scanty
1-9 AFB/1 length or 100 HPF
1-19 AFB/1 length
1-29 AFB/1 length
1+
10-99 AFB/1 length or 100 HPF
20-199 AFB/1 length
30-299 AFB/1 length
2+
1-10 AFB/1 HPF on average
5-50 AFB/1 HPF on average
10-100 AFB/1 field on average
3+
>10 AFB/1 HPF on average
>50 AFB/1 HPF on average
>100 AFB/1 field on average
Abbr: AFB: Acid Fast Bacilli; HPF: High Power Field
Resources
- Training Module (1-4) for Programme Managers and Medical Officers, NTEP, MoHFW, 2020.
- Manual for Sputum Smear Fluorescence Microscopy, RNTCP, MoHFW, 2007.
- Fading of Auramine-stained Mycobacterial Smears and Implications for External Quality Assurance.
Assessment
Question
Answer 1
Answer 2
Answer 3
Answer 4
Correct answer
Correct explanation
Page id
Part of Pre-test
Part of Post-test
Immersion oil is used to view fluorescent-stained smears.
True
False
2
Immersion oil is not required to view fluorescent-stained smears as they are observed with 20x or 40x objective lens.
Yes Yes Reporting and Recording results of Smear Microscopy (On paper)
ContentEach Designated Microscopic Center (DMC) needs to maintain a National TB Elimination Program TB (NTEP) Laboratory Register which collects information of all presumptive TB patients who undergo tests or confirmed patients who undergo follow-up tests.
- After doing the Acid-fast Bacilli (AFB) test, the laboratory technician needs to ensure that all the microscopy test results are entered against the name and the serial number of the presumptive/ confirmed TB patients.
- The result needs to be mentioned in terms of positive or negative test results along with the AFB grading.
- It is expected that the positive results are written in RED and negative in BLUE point pens.
- The AFB results can be mentioned for newly diagnosed TB patients for both spot as well as morning sample and must include the Nikshay ID of the patient as well as the test date and result.
- In case, the sample is repeated, the same is also mentioned.
- All the information also needs to be entered into NTEP Nikshay online application using a tablet or mobile phone.
- In a setting where Nucleic Acid Amplification Test (NAAT) is co-located, another sample needs to be tested for Rifampicin (Rif)-resistance and the same result also needs to be noted into the TB laboratory register.
- The performance of each laboratory is also entered into the monthly DMC extract called Annexure M which is consolidated for all DMCs by the concerned Senior TB Laboratory Supervisor (STLS).
- HIV and diabetes tests are also offered to each confirmed TB case and the details of the same are also part of the TB laboratory register.
- Patients who have been tested also need to be provided with the test results in a hard copy apart from the provision of information to the treating clinician of the health facility.
Resources
Kindly provide your valuable feedback on the page to the link provided HERE
Reporting results of Microscopy (Digitally)
ContentResults of Ziehl–Neelsen (ZN) and Fluorescence Microscopy (FM) are added to Nikshay Diagnostic Module. After a patient is registered, test details are added which then leads to the page to add details on sample type, facility and test results (Figures 1-6).
Features of Nikshay Diagnostic Module
- A simplified user workflow for adding and updating test requests and results.
- Tests can be added and all the added tests’ history is available.
- Ability to map samples to one or more tests within Nikshay.
- A “Workbench” view is available to provide a single interface to view/ print/ copy the Test Summary, Add/ View Sample Details and Update/ View Result details.

Figure 1: Nikshay Screenshot to Add Tests under Diagnostics Module.
To add results for ZN/FM, the test types either Microscopy ZN or Microscopy Fluorescence should be selected from the drop-down menu (Figure 2).

Figure 2: Nikshay Screenshot to Add Reasons for Test under Diagnostics Module.
In case of follow-up with ZN/FM Microscopy, the reason for follow-up should be selected (Figure 3).

Figure 3: Nikshay Screenshot to add Follow-up ZN/FM Microscopy Test under Diagnostics Module.

Figure 4: Nikshay Screenshot to add Test Type for ZN/FM Microscopy and Facility Details under Diagnostics Module.
Details on sample type and sample description (mucopurulent/ blood-stained/ saliva) are added (Figure 5).

Figure 5: Nikshay Screenshot to add Sample Details and Description for ZN/FM Microscopy under Diagnostics Module.
Under test results, as shown in Figure 6, details of lab serial number, sample number, test date, test reported date and final interpretation of results are added based on the selection from a drop-down menu that includes options for:
- Negative
- Positive (1+, 2+, 3+)
- Positive (Scanty 1-9)

Figure 6: Nikshay Screenshot to add Test Results for ZN/FM Microscopy under Diagnostics Module.
Resources
Assessment
Question
Answer 1
Answer 2
Answer 3
Answer 4
Correct answer
Correct explanation
Page id
Part of Pre-test
Part of Post-test
What sample descriptions are added in Nikshay for ZN/FM Microscopy under Diagnostics Module?
Mucopurulent
Blood-stained
Saliva
All of the above
4
Sample descriptions (mucopurulent/ blood-stained/ saliva) are added in Nikshay for ZN/FM Microscopy under Diagnostics Module.
Yes
Yes
Generating the DMC Laboratory Register from Nikshay
ContentThe DMC lab register can be generated from Nikshay as a spreadsheet and stored electronically or printed for paper records.
Steps for generating DMC Register from Nikshay:
- Login to Nikshay Reports using your login ID and password.
- Under Reports, click on ‘Patient wise List’ and select the option for ‘DMC Register’ (see screenshot below)


Figure: Screenshot of Nikshay Reports to generate DMC Register
A few filters need to be selected to generate the register.
- First is the geographic location (State/ District/ TU/PHI); most of these are preselected.
- This is followed by a selection of the period for which the register has to be generated. This includes selection of ‘Frequency’ (either monthly or annually)of the report and the month or quarter for which the report is required.
- Click on the ‘Generate Excel’ button and it will lead you to a page from which you can download the register.
Things to remember:
- You can generate a DMC register for facilities below your login level (e.g., a district can generate a DMC register of a TB Unit or DMC of the same district. and a State can generate a register for any District under it)
- The data included in the register is based on the "Date reported" of a test.
Video fileVideo: Steps to access the DMC Register in Ni-kshay
Resource
Assessment
Question Answer 1 Answer 2 Answer 3 Answer 4 Correct answer Correct explanation Page id Part of Pre-test Part of Post-test Where do you find the option to generate a DMC Register? In Nikshay under the "Admin" tab In Nikshay under the Others tab In Nikshay Reports under the "Patient wise List" tab In Nikshay Reports under the "Reports" tab 3 The option to generate a DMC Register is available under the Nikshay reports tab under the "Patient wise List" tab. Yes Yes Tuberculosis Laboratory Register
ContentThe Tuberculosis (TB) Laboratory Register is a paper-based recording register kept in all National TB Elimination Programme (NTEP) laboratories for recording details of diagnostic services offered to TB patients referred from both private and public health facilities.
The register is maintained in the Designated Microscopy Centre (DMC). It is the only register used for recording the details of specimen smear examinations. The Laboratory Technician (LT) is responsible for maintaining and updating the laboratory register.
There are two portions in the TB lab register and the table below shows the details captured in each portion.
Table: Pages and Information Covered in the TB Laboratory Register; Source: NTEP Training Module 2 for Programme Managers & Medical Officers
PORTION
DETAILS CONTAINED
Left-hand Portion
Lab serial number assigned by the LT
Date of the sample collection
Patient details such as name, sex, age, address, and contact number
Information on if the patient is from a key population
Referring health facility details
Reasons for examination
Right-hand Portion
Details on the type of specimen
Visual appearance results along with dates
Comorbidity status (HIV and Diabetes)
Details of drug susceptibility testing
Nikshay ID / Notification
Treatment initiation details
The last two columns are for the LT’s signature and remarks by the LT or supervisor
Important Points to Note
- Duplicate registers should not be maintained.
- LTs should ensure that the correct laboratory serial number is recorded.
- Laboratory serial number is given to the patient and not to the sample. A new number should be assigned to every presumptive TB case whose sputum is to be examined.
- All smear-positive (including scanty) results should be recorded in red ink.
- No over writing or manipulation in already entered data should be done.
The figure below shows the left-hand portion of the TB lab register. Click here to access the full form in the NTEP Training Module 2 for Programme Managers & Medical Officers, p. 219.
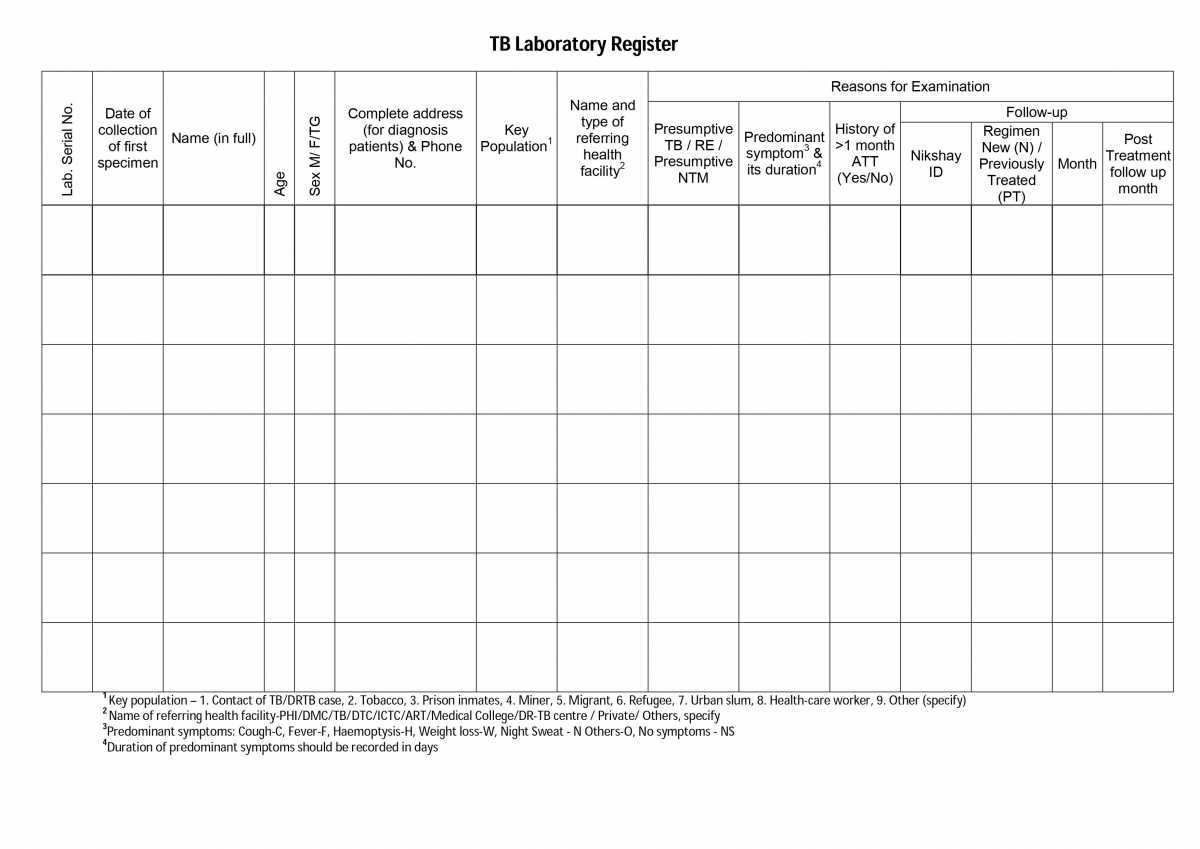
Figure: First Page of the TB Laboratory Register; Source: NTEP Training Modules 1-4 for Programme Managers & Medical Officers, p. 219
Resources
Kindly provide your valuable feedback on the page to the link provided HERE
-
CDST_LT: Maintenance of the microscope
FullscreenCleaning a Microscope
ContentRegular cleaning of microscope is essential to ensure that it is optimally functioning. Cleaning is important to remove dirt, lens immersion oil and ensure disinfection. It involves cleaning the lenses, the body, the stage of the microscope and the light source. The microscope needs to be cleaned daily and the lenses need to be cleaned after each use.
Materials used for cleaning:
- Lint free cloth
- Lens paper
- Lens cleaning solution
- 70% ethanol
- Detergent solution
Procedure for Cleaning Different Parts of the Microscope
- The eyepiece shades, stage, focusing knob and nose piece are commonly touched during microscope operation, so these parts and the body of the microscope must be cleaned first to remove any stains with a neutral detergent /solution recommended by the manufacturer.
- To clean the microscope eyepiece, objective lens, surfaces of the condenser and the light exit glass, moisten a lens paper with 1 to 2 drops of lens cleaning solution and clean the lens/ glass with a circular/spiral motion (as shown in the figure below).
- Dry with a clean, dry piece of lens paper.
- Use the Spiral Wiping Technique: Wipe from the center to the periphery in a circular motion.
- The oil immersion lens should be cleaned after each use to ensure that immersion oil is not left on the surface.
- The microscope surface and parts (except the lenses) need to be disinfected using 70% ethanol.

Figure: Cleaning of Lenses using the Spiral Wiping Technique. Source: How to Clean the Microscope
Important Considerations for Cleaning Microscopes
- Do not blow air to remove the dirt.
- Do not wipe the lens with an ordinary cloth. All the lenses should be cleaned with dry lens paper/lint-free cloth.
- Never use spirit or alcohol or xylene to clean the lenses as these can damage them.
- Avoid using organic solvents that may damage plastic parts.
- Whenever possible, use the cleaning fluid recommended by the manufacturer.
Video fileResources
- Laboratory Diagnosis of Tuberculosis by Sputum Smear Microscopy, GLI, 2013.
- Module for Laboratory Technicians, CTD, 2005.
- How to Clean and Sterilise Your Microscope, Olympus, Life Science Solutions, 2020.
Assessment
Question
Answer 1
Answer 2
Answer 3
Answer 4
Correct answer
Correct explanation
Page id
Part of Pre-test
Part of Post-test
The lenses of the microscope should be cleaned with 70% alcoholic solution.
True
False
2
Lenses should never be cleaned with alcoholic solution and should always be cleaned with lens solution and lens paper.
Yes
Yes
At what frequency should the lenses of a microscope be cleaned? After each use Daily Weekly Monthly 1 The oil immersion lens should be cleaned after each use to ensure that the immersion oil is not left on the surface. Yes Yes Storing a Microscope
ContentProper handling, maintenance and storage of the microscope are essential for proper functioning and life of the microscope.
The microscope should be placed and stored preferably in a box in a dry, dust-free and vibration-free environment, which is specially built in the laboratory (as shown in the figure below).

Figure: Storage of microscope in a wooden box; Source: Laboratory diagnosis by sputum smear microscopy
Important considerations for the storage of microscope
- When the microscope is not being used/ stored overnight, it should be covered or kept in a storage cupboard/box to keep it free from dust.
- Avoid exposing the microscope to direct sunlight.
- Avoid exposing the microscope to moisture as humidity may allow fungus to grow on the lens and cause rusting of the metal parts.
- The stored microscope must be kept warm with a light source to prevent growth of fungus. A bulb holder for a 15-watt bulb should be fixed on the rear wall of the storage cupboard, such that the microscope does not contact the bulb while storing or removing the microscope. The bulb should remain on when the microscope is stored inside the box
- An alternative may be to place plenty of dry blue silica gel into a shallow plate and place it in the box when the microscope is kept in it. Silica gel is blue in colour when it is dry, but when it becomes wet /absorbs moisture it turns pinkish. As soon as the silica gel becomes pink, it should be changed or heated until it turns blue again and then be reused.
Resources
Assessment
Question
Answer 1
Answer 2
Answer 3
Answer 4
Correct answer
Correct explanation
Page id
Part of Pre-test
Part of Post-test
Silica gel is blue in colour when it is dry.
True
False
1
Silica gel is blue in colour when it is dry.
Yes
Yes
Common issues with Microscope that require technical support
ContentRemovable parts in a microscope that need replacement include objectives, eyepieces, light bulbs, fuses. In case of repair/ technical support, only competent agency that handles maintenance of instruments should be contacted. Laboratory personnel should never attempt to dismantle any part of the microscope for repair.
Some of the common technical problems encountered, their likely causes and solutions are given in the table below.
Problems
Likely causes
Solutions
The viewing field is too dim
Condenser is too low
Condenser iris diaphragm is closedRaise condenser to correct its position
Open the diaphragm properlyDark shadows in the field that move as you turn the eyepiece
Surface of eyepiece is scratched
Eyepiece is dirtyReplace the eyepiece
Clean the eyepieceImage with the high-power objective is not clear
Slide is upside down
There is an air bubble in the oil
There is dirt on the objectiveTurn the slide over.
Move 100x lens quickly from side to side
Clean the lensImage with the low-power objective is not clear
There is oil on the lens
There is a layer of dust on the upper surface of the objectiveClean the lens
Mechanical stage is not moving, too stiff or does not stay up
Poor tension adjustment on the mechanical stage
Solidified lubricantsAdjust tension with adjustment knob
Needs serviceIn case, the viewing field is still dim and cloudy, consider the following possible causes and contact the competent personnel for cleaning/ repair:
- Massive growth of fungus on the lenses or prisms due to storage in a high-humidity environment
- Penetration of immersion oil between the lenses of the objective
- A damaged objective (as a result of careless focusing, dropping, rough changing of slides).
Video fileResources
1.
Module for Laboratory Technicians
2.
Laboratory diagnosis by sputum smear microscopy
Assessment
Question
Answer 1
Answer 2
Answer 3
Answer 4
Correct answer
Correct explanation
Page id
Part of Pre-test
Part of Post-test
What is the possible cause for dark shadows in the field?
The scratched eyepiece or dirty eyepiece
Broken lens
Solidified lubricants
Stage stuck
1
Dark shadows in field appear due to a scratched eyepiece or dirty eyepiece.
Yes
Yes
Annual Maintenance [AMC] of a Microscope
ContentThe binocular microscope requires considerable care in its use, regular cleaning, and protection from dust and fungal growth.
Annual maintenance of a microscope includes:
- Checking for broken or damaged parts and ensuring that the lenses, mirrors and other light-conducting surfaces are clean.
- Cleaning the lenses first for dirt with a blower brush, then wiping with lens solution on a lens paper/ 70% alcohol on the lens paper.
- Removing eyepieces or objectives from their fixation holes for cleaning.
- Properly cleaning the lower lens of the condenser after removal from its fixing.
- Cleaning the slide holder after removing from the mechanical stage.
- Cleaning for any deposition of immersion oil and fungal growth on the prisms in the binocular tube and eyepieces
- Changing electrical services including bulbs and fuses.
- Greasing/ lubrication of movable parts.
National Tuberculosis Elimination Programme (NTEP) has earmarked INR 2000 for Annual Maintenance [AMC] per binocular microscope including spare parts and repairs.
Resources
- Module for Laboratory Technicians.
- Laboratory diagnosis by sputum smear microscopy.
- Norms and Basis of Costing.
Assessment
Question
Answer 1
Answer 2
Answer 3
Answer 4
Correct answer
Correct explanation
Page id
Part of Pre-test
Part of Post-test
What is the AMC cost for each binocular microscope under NTEP?
INR 1000
INR 5000
INR 3000
INR 2000
4
NTEP has earmarked INR 2000 for Annual Maintenance (AMC) per binocular microscope, including spare parts and repairs.
Yes
Yes
-
CDST_LT: Maintaining supplies of lab consumables at the DMC
FullscreenConsumables required at a DMC
Content- Designated Microscopy Centres (DMCs) are the most peripheral laboratory under the National TB Elimination Programme (NTEP) network.
- Any person identified to be a presumptive TB patient is first referred to the nearest DMC for sputum examination or their sputum specimens are collected and transported to the DMC. Therefore, it is very important for the DMCs to maintain an adequate stock of reagents and other consumables.
- The Medical Officer (MO) of DMC is responsible for determining the number of reagents and other materials the DMC needs every month.
- The Senior Tuberculosis Laboratory Supervisor (STLS) ensures these supplies are distributed in a timely manner, as and when required.
- The Lab Technician (LT) is responsible for exhausting the old supplies before the new ones.
The consumables required at the DMC can be broadly categorised as:
- Consumables required for sputum collection
- Consumables required for slide preparation
- Consumables required for smear examination
- Consumables stationery
- Other
Table: List of Items with Technical Specifications of Laboratory Consumables Required for DMCs Sl. No. Name of the item Technical Specifications - For Sputum collection
Sputum containers Cups made of Special Medical Grade Polypropylene, thin plastic, translucent, diameter - 4 cm, capacity - 30 ml, the screwable cap should also be made of Special Medical Grade Polypropylene and should be airtight and leak-proof. Absorbent cotton 500 gms/roll Phenolic compound 5% phenol/ 40% phenolic compound (proprietary Phenyl) diluted to 5% Adhesive labels for sputum containers Sputum specimen transport box Insulated box, made of plastic 10” x 10” x 10”, thickness 1” with lid, handle and nylon belt 1” width 2.5 feet length, nylon strap of 1” width 2 feet length with velcro to strap the lid of the box from side to side. - For slide preparation
Slides for Microscopes Glass slides plain, size = 76mm x 26mm x 1.3mm, clean, scratch-free with smooth edges, uniform refractive index, pack of 50. Broomstick 10 cms in length and thick enough to make good smears. Diamond marker pencil 6" (15.24 cm.) holder with artificial diamond (hard stone) embedded at one end with a screw cap, to mark on microscope glass slides. Grease marking pencil Marking pencil MPS, blue or red coloured, 8" length, to write on glassware/ metal surfaces. Staining racks For drying the slides. Slide boxes for storing slides Glass (or metal) rods For holding slides during the staining process. Forceps, Chitel forceps Stainless steel for slides, 15 cm. Scissors 25 cm, stainless steel Ziehl Neelsen stain Auramine O fluorescent dye for Fluorescence Microscopy (FM) Wherever FM is being done Whatman filter paper No 1 3 ) For smear examination Binocular Microscope With 10x, 40x and oil immersion objective (100x) eyepieces (10x) and spare bulbs and fuses. Immersion oil Filter paper To drain the oil from the slides. Fine Silk and Lint cloth Lens paper For wiping the oil immersion lens after examination of each slide 4) Consumables - Stationery Request form for examination of biological specimen for sputum examination TB Laboratory Register Referral/ Transfer form for treatment Stock register – laboratory Marker pen 5 ) Other Plastic tumblers/ mugs Time (stop-watch) Spirit lamp or Bunsen burner Silica gel Hygroscopic agent to maintain the microscope in a moisture-free environment (to be placed in the cabinet for Binocular Microscope). Alcohol (absolute) Ethanol Cotton, full sleeves Aprons Disposable gloves 6 and 8 inches Methylated spirit Resources
- Training Module (1-4) for Program Managers and Medical Officers, NTEP, MoHFW, 2020.
- Guidelines for Quality Assurance of Smear Microscopy for Diagnosing Tuberculosis, RNTCP Lab Network, CTD, MoHFW, India, 2005.
- Module for Laboratory Technicians, CTD, MoHFW, India, 2005.
Assessment
Question Answer 1 Answer 2 Answer 3 Answer 4 Correct answer Correct explanation Page id Part of Pre-test Part of Post-test Sputum containers and their screwable cap used for sputum collection at the DMC should be made of Special Medical Grade Polypropylene and should be air-tight. True False 1 Sputum containers and their screwable cap used for sputum collection at the DMC should be made of Special Medical Grade Polypropylene and should be air-tight and leakproof. Yes Yes Who ensures that supplies are distributed in a timely manner at DMCs? Treatment Supporter Medical Officer Senior Tuberculosis Laboratory Supervisor (STLS) Lab Technician (LT) 3 The Senior Tuberculosis Laboratory Supervisor (STLS) ensures these supplies are distributed in a timely manner, as and when required.
Yes Yes Stock Register at a DMC
Content- Designated Microscopy Centres (DMCs) are the most peripheral laboratory under the National TB Elimination Programme (NTEP) network. Therefore, it is very important for the DMCs to maintain an adequate stock of all consumables.
- A paper-based stock register is maintained at the DMC and submitted as a part of the ‘Monthly report on programme management, logistics and microscopy’.
- All the DMCs as well as Peripheral Health Institute (PHI) that are a DMC need to fill the second part of this monthly report format for reporting the status of laboratory consumables and equipment (Figure below).

Figure: Monthly report format for reporting status of laboratory consumables and equipment, to be filled by PHI that is DMC.
- The stock register for consumables at the DMC has the provision to enter the information about the stock of consumables that are available at the DMC on the first day of the month, stock received and consumed during the month and stock remaining on the last day of the month along with the requested quantity of new stock.
- The Lab Technician (LT) of the DMC is responsible for exhausting the old supplies before the new ones.
- The Medical Officer (MO) of the DMC is responsible for determining the stocks and the Senior Tuberculosis Lab Supervisor (STLS) should ensure these supplies are distributed in a timely manner, as and when required.
Table: Calculation of Stocks Required at the DMC
Sputum containers For diagnosis:
- During the first week of each quarter, the number of new smear-positive cases registered and treated during the last quarter should be determined and this number should be multiplied by 10.
- Ten is the average number of symptomatic required to be examined for detecting one case of new pulmonary smear-positive tuberculosis.
- Since two sputum specimens are taken for each symptomatic patient, further multiply the number obtained above by 2.
For follow-up:
- Two follow-up specimens are taken for the majority of patients - one at the end of the intensive phase and the other at the end of treatment. One sputum container is needed for each follow-up.
- Once the number of sputum containers required has been calculated, allow sufficient reserve stock for three months, add 10% to account for wastage of sputum containers, and account for the sputum containers in stock.
- On the last working day of the quarter, count the number of sputum containers in stock and subtract from that needed for diagnosis and follow-up examinations as calculated above.
Slides - Once the number of sputum containers needed for the next quarter is determined, order a slightly higher number of slides than the required no. of sputum containers to account for unavoidable breakage of slides.
Reagents - Reagents are supplied to the DMCs on monthly basis by the District TB Centre (DTC).
- The stock register should have the mention of expiry dates of the reagents and the first expiring reagents must be exhausted first based on the First Expiry First Out (FEFO) principle.
- The reagents should not be used beyond three months from the date of preparation.
CBNAAT/ Truenat Machines and Cartridges/ Chips
- These are procured centrally and supplied to state/ district/ sites based on their requirement.
- Recording, reporting and monitoring of cartridges/ chips is done through Nikshay-Aushadhi and they are supplied based on the stock availability, consumption and expected case load.
Binocular Microscopes (BM) and LED Fluorescence Microscopes (FM) - 1 BM is required for every DMC.
- The no. of BM in place and no. of BM in working condition should be reported in the stock register.
- LED FM is supplied to the high case load facilities where the workload is more than 25 slides per day.
Tuberculosis Laboratory Register - Each TB lab register allows for the registration of at least 2000 patients.
- For each lakh population, 75 smear-positive patients are projected, requiring the examination of 750 patients (thrice each). Additional follow-up examinations will bring the number of registers needed to approximately one lab register/ lakh.
- If there are blank pages in the register at the end of a year, it can be used the following year. However, every year should be started on a new page.
Laboratory Form for Sputum Examination - For diagnosis, approximately 10 laboratory forms for sputum examination are needed. (10 is the average number of symptomatic for each case of pulmonary smear-positive tuberculosis identified).
- For follow-up, approximately 0.2 laboratory forms for sputum examination are needed for each pulmonary tuberculosis case. (1 out of 10 examined will be smear-positive, each needs two forms for follow-up. When calculated, out of 10 it will be 0.2)
Resources
- Training Modules (1-4) for Programme Managers and Medical Officers, CTD, MoHFW, GoI, 2020.
- Module for STS Part 2: Ensuring Proper Registration and Reporting. CTD, MoHFW, India.
Assessment
Question Answer 1 Answer 2 Answer 3 Answer 4 Correct answer Correct explanation Page id Part of Pre-test Part of Post-test How many TB Laboratory Register/s is/are required in one year for each DMC? 1 2 3 4 1 Each Tuberculosis Laboratory Register allows for the registration of at least 2000 patients. For each lakh, 75 smear-positive patients are projected, requiring the examination of 750 patients (thrice each). Additional follow-up examinations will bring the number of registers needed to approximately one lab register/ DMC. Yes Yes For follow-up, approximately 0.2 laboratory forms for sputum examination are needed for each pulmonary tuberculosis case.
True False 1 For diagnosis, approximately 10 laboratory forms for sputum examination are needed. 10 is the average number of symptomatic for each case of pulmonary smear-positive tuberculosis identified. 1 out of 10 examined will be smear-positive, each needs two forms for follow-up. When calculated, out of 10 it will be 0.2
Yes
Yes
Indenting Supplies at a DMC
ContentThe Medical Officer (MO) of Designated Microscopy Centre (DMC) is responsible for determining the amount of reagents and other materials the DMC needs every month.
The Senior TB Laboratory Supervisor (STLS) will make sure these supplies are distributed in a timely manner, usually on a monthly basis or as and when required.
- STLS place an order for DMC reagents and consumable four times a year based on the approximate number of sputum specimens the laboratory examined in the previous quarter.
- The STLS should order the sputum containers during the first week of the quarter so that the health units and microscopy laboratories have enough sputum containers to collect sputum specimens and the microscopy laboratories have enough slides to conduct sputum smear examinations.
The calculation of reagents/ consumables required for examination of 3000 smears is shown in Table 1.
Reagents/ Equipment for staining
Quantity
Binocular microscope with 10x, 40x and oil immersion objective (100x) eyepieces (10x) and spare bulbs and fuses
At least 1 per DMC
Plastic disposable sputum containers
3,300
Slides for microscope, 25*75 mm, 1.1 mm-1.3 mm Thick
3,300
Broom stick 10 cms length
3,300
Diamond marker pencil
3 number
Timer, 30 or 60 minutes
1 number
Forceps, Chitel forceps stainless steel for slides 15 cm
1 number
Scissors, 25 cm stainless steel
1 number
Slide rack, Staining slide rod of metal or plastic or glass for 12 slides
2 numbers
Slide boxes, For 100 slides
33 boxes + 2 per DMC for RBRC
Tissue rolls
4 numbers
Marker pen
12
Absorbent cotton, 500 gms/ roll
4 numbers (2 k.g)
Pressure cooker, For disposal by autoclaving
Optional
Phenol with concentration mentioned on the bottle
80 litres
Methylated spirit
3 litres
Cotton, full sleeves aprons
2
Disposable gloves, 6 and 8 inches (box of 25 pairs)
12 boxes
Spirit lamp with wicks
1 number
Metal wire, for swab for heating Carbol fuchsin
1 number
Sputum specimen transport box, Insulated box, made of plastic 10” x 10” x 10”, thickness 1” with lid, handle and nylon belt 1” width 2.5 feet length, nylon strap of 1” width 2 feet length with Velcro to strap the lid of the box from side to side.
2 numbers
Table 1: Calculation of Consumables Required at DMC for Examination of 3000 Smears; Source: Training Modules for Programme Managers and Medical Officers
- The monthly report on Programme Management, Logistics and Microscopy at the Peripheral Health Institution (PHI) level are filled by DMCs which are PHI as shown in Table 2.
Laboratory Consumables (To be filled in by only PHIs which are a DMC)
Item
Unit of Measurement
Stock on first day of Month
Stock received during Month
Consumption during Month
Stock on last day of Month
Quantity requested
Sputum containers*
Nos.
Universal containers for C & DST
Nos
Slides
Nos.
Carbol Fuchsin (1% solution)
Litres
Methylene Blue (0.1% solution)
Litres
Sulphuric Acid (25% solution)
Litres
Phenolic solution(for disinfection-~40% pure solution)
Litres
Immersion oil/Liquid Paraffin (Heavy)
mL
Methylated Spirit
Litres
* PHIs that are not a DMC, but have been supplied with sputum containers, should complete this row.
Table 2: Request for Laboratory Consumables and Reagents in monthly reports from PHI which are DMCs; Source: Monthly Report on Programme Management, Logistics and Microscopy Peripheral Health Institution Level
- The DMC reagents and consumables are budgeted in the District Annual Action Plan under laboratory materials as per the amount permissible based on District Performance in the last four quarters.
- Procurement planned during a financial year is based on last year’s specimen processed/ slides examined and amount spent for purchase of lab material for smear microscopy.
Resources
- Training Modules for Programme Managers and Medical Officers.
- Monthly Report on Programme Management, Logistics and Microscopy
Peripheral Health Institution Level (https://tbcindia.gov.in/showfile.php?lid=3055)
Assessment
Question
Answer 1
Answer 2
Answer 3
Answer 4
Correct answer
Correct explanation
Page id
Part of Pre-test
Part of Post-test
The MO of the DMC is responsible for determining the amount of reagents and other materials the DMC needs every month.
True
False
1
The MO of DMC is responsible for determining the amount of reagents and other materials the DMC needs every month.
Yes
Yes
Preparation of Reagents for ZN Microscopy
ContentThe National TB Elimination Program (NTEP) recommends freshly preparing reagents from commercial procured products, at the District TB units to be used for microscopy. Chemicals need to be carefully monitored for potency and the calculation needs to be accurate while preparing reagents for smear microscopy.
For Preparing 1% Carbol Fuchsin (500ml): The required chemicals and materials for preparing 500 ml Carbol Fuchsin are:
1. Basic Fuchsin
- Chemical name: Pararosaniline hydrochloride
- Chemical structure: C19H18N3Cl
- Molecular Wt: 323.8
- Colour: Metallic green
Potency correction factor: Note down the dye content – this should be available on the container. The dye content should be approximately 85% - 88%. To calculate the required amount of basic fuchsin, divide the actual amount required by the dye content. For example: dye content = 85%, actual amount required = 5gms, required amount of dye = 5/0.85 = 5.88 gms.
2. Ethyl Alcohol: 50 ml (Absolute alcohol, purity must be 98-100%)
3. Carbolic acid crystals (Phenol): 25 gms
- Chemical name: Phenol
- Chemical structure: C6H5OH
- Molecular Wt: 94.11
- Melting point: 40°C+2.5
4. Distilled purified water: 500 ml
Steps for Preparing 1% Carbol Fuchsin (500ml)
- Add 25 gms of phenolic crystals to a conical flask
- Add 50ml ethanol
- Mix well until the crystals completely dissolves. Add 50 ml distilled water if required.
- Conical flask should be kept in water bath set at 60°C or in a trough containing warm water
- Weigh required amount of basic fuchsin powder and transfer it into a conical flask
- Mix well until the crystals dissolve well.
- Make the total volume to 500ml by adding distilled water
- Filter using Whattman filter paper and transfer into a bottle
- Label as 1% Carbol Fuchsin - Primary stain
- Date of preparation, date of expiry, batch no. and the name of the technician or STLS who has prepared the stain should be clearly mentioned on the bottle.
- Store it in cool place, away from direct sunlight
- Any time particles start to form in the Carbol Fuchsin solution, the solution must be filtered again
For Preparing 25% Sulphuric Acid (500ml):
- Chemical structure: H2SO4
- Molecular wt: 98.08
- Purity: 95-97%
- Colour: Clear
- Measure 375ml distil water into a 1L flask
- In a glass cylinder, measure 125ml concentrated Sulphuric Acid. Pour it slowly and gently into the conical flask containing distilled water. Note: Always add acid to water. Never add water to acid
- To dissipate the heat generated, place the flask in a trough of water
- Mix well
- Allow to cool
- Transfer into a bottle
- Label as 25% Sulphuric Acid - Decolorizing Solution
- Date of preparation, date of expiry, batch no. and the name of the technician or STLS who has prepared the solution should be clearly mentioned on the bottle.
For preparing 0.1% Methylene Blue (500ml):
- Chemical name: Methylthionine chloride
- Chemical structure: C16H18ClN3S
- Molecular Wt: 319.9
Potency correction factor: Note down the dye content – this should be available on the container. The dye content should be approximately 82%. To calculate the required amount of methylene blue, divide the actual amount by the dye content. For example: dye content = 82%, actual amount required = 0.5gms, required amount of dye = 0.5/0.82 = 0.61 gms.
- Weigh the required amount of Methylene Blue
- Dissolve it in 500ml distilled water
- Transfer into a bottle
- Label as 0.1% Methylene Blue
- Date of preparation, date of expiry, batch no. and the name of the person who prepared the solution should be clearly written on the bottle.
Table: Preparation of different volumes of stains for ZN microscopy
Ziehl-Neelsen method
Quantity of reagent for 5L
Quantity of reagent for 1L
Quantity of reagent for 500 ml
Quantity of regent for 100 ml
Basic fuchsin (dye content/purity if 85%)
58.8 g
11.76 g
5.88 g
1.17 g
Alcohol
500 ml
100 ml
50 ml
10 ml
Phenol crystals
250 g
50 g
25 g
5 g
Distilled water
To make final volume 5000 ml
To make final volume 1000 ml
To make final volume 500 ml
To make final volume 100 ml
Carbol-fuchsin 1%
Sulfuric acid
1250 ml
250 ml
125 ml
25 ml
Distilled water
3750 ml
750 ml
375 ml
75 ml
H2SO4 25%
Methylene blue (dye content if 82%)
6.1 g
1.22 g
0.61 g
0.12 g
Distilled water
5000 ml
1000 ml
500 ml
100 ml
Methylene blue 0.1%
Always perform a quality check of the reagents prepared by using Quality Control Positive and Quality Control Negative. If purity is not available on the reagent bottle, then search the website of the company of which the reagent was procured or ask the company about the certificate mentioning about the purity of reagent
Please watch the video below for more information:
Video fileResources
- Module for Laboratory Technicians (RNTCP), Central TB Division, MoHFW, 2005
- TB Lab Consumables Specifications 2019
- SoP for preparation of Laboratory Reagents - WHO
- Laboratory Diagnosis of Tuberculosis by Sputum Microscopy, GLI Initiative
Kindly provide your valuable feedback on the page to the link provided HERE
Preparation of Reagents for FM Microscopy
ContentReagent preparation is an important activity and it’s essential to use certified chemicals and reagents. One should always check the potency of the chemicals used and calculate the amount to be weighed accordingly.
The following are the steps for reagent (primary stain and counter stain) preparation for Florescence Microscopy:
0.1% Auramine–O (1 L), the primary stain
- Weigh 1 gm Auramine–O and transfer to conical flask.
- Add 30 gm Phenolic crystals (99.5% purity) and mix well.
- Add 100 ml absolute Ethanol (98-100% purity) and mix well.
- Add 870 ml distilled water to make up final volume of 1 L.
- Filter and transfer to amber bottle.
- Label the bottle as 0.1% Auramine–O.
- Date of preparation, date of expiry, batch no. and name of the Senior TB Lab Supervisor (STLS) who has prepared the stain should be clearly mentioned on the bottle.
1% Acid Alcohol (1 L) to decolorize
- Dissolve 5 gm Sodium chloride in 250 ml distilled water.
- Add 5 ml concentrated hydrochloric acid.
- Mix with 750 ml absolute alcohol.
- Always add acid slowly to alcohol, not vice-versa.
- Store in amber coloured bottle.
- Label the bottle as 1% Acid Alcohol.
- Date of preparation, date of expiry, batch no. and name of the STLS who has prepared the stain should be clearly mentioned on the bottle.
0.5% Potassium Permanganate, KMnO₄ (1 L), the counter stain
- Weigh 5 gm of Potassium permanganate (99.5% purity).
- Transfer to a conical flask.
- Take 1 L of distilled water.
- Add small amounts of distilled water tothe conical flask containing KMnO₄ and mix well.
- Add the remaining distilled water to make up final volume of 1 L.
- Filter and transfer to a bottle.
- Label the bottle as 0.5% Potassium permanganate.
- Date of preparation, date of expiry, batch no. and name of the STLS who has prepared the stain should be clearly mentioned on the bottle.
Table: Preparation of Different Volumes of Stains for FM Microscopy
AURAMINE METHOD
QUANTITY OF REAGENT FOR 5 L
QUANTITY OF REAGENT FOR 1 L
QUANTITY OF REAGENT FOR 500 ML
QUANTITY OF REAGENT FOR 100 ML
Auramine
Ethanol
Phenol
Distilled water
0.1% Auramine
5.0 g
500 ml
150.0 g
To make final volume 5000 ml
1.0 g
100 ml
30.0 g
To make final volume 1000 ml
0.5 g
50 ml
15.0 g
To make final volume 500 ml
0.1 g
10 ml
3.0 g
To make final volume 100 ml
Hydrochloric acid
Sodium chloride
Ethanol
Distilled water
1% Acid–alcohol
25 ml
25.0 g
3750 ml
1250 ml
5 ml
5.0 g
750 ml
250 ml
2.5 ml
2.5 g
375 ml
125 ml
0.5 ml
0.5 g
75 ml
25 ml
Potassium permanganate
Distilled water
0.1% Potassium permanganate
25.0 g
5000 ml
5.0 g
1000 ml
2.5 g
500 ml
0.5 g
100 ml
Video fileResources
Kindly provide your valuable feedback on the page to the link provided HERE
Fullscreen