-
CDST_LT: Diagnostic technologies in NTEP
FullscreenMicroscopy
ContentMicroscopy is a TB diagnostic technology that utilizes the acid-fastness property of Mycobacterium tuberculosis to visualize it under a microscope. Results of sputum smear microscopy can either be smear-negative, or smear-positive (with various grades).
Advantages:
- It is currently the most accessible and cheapest TB diagnostic test available under National TB Elimination Programme (NTEP) in India.
- It has the shortest turnaround time for diagnosis.
- It has high specificity.
Limitations:
- Low sensitivity. It becomes positive only when more than 5000 bacilli/ml of sample are present. Hence, cases would be missed in early disease, or when an inappropriate biological specimen is provided, where bacterial load in sputum is less.
- It is unable to differentiate between M. tuberculosis and Non-tuberculous Mycobacteria (NTM). This is predominantly an issue in geographies with lower burden.
There are two types of microscopies used in NTEP: Ziehl-Neelsen (ZN) Microscopy and Fluorescence Microscopy (FM). These vary in the type of stain and microscope used. FM is newer of the two types and is currently recommended for use over ZN.
Resources
Ziehl–Neelsen Microscopy
ContentIn Ziehl-Neelsen microscopy, the carbol fuchsin fuchsin stain is heated to enable the dye to penetrate and bind the waxy mycobacterial cell wall. Following acid-decolourisation, the sputum smear is counterstained with methylene blue which stains the background material, providing a contrast blue colour against which the red AFB can be seen.
On observation under a microscope with oil immersion at 100X magnification, AFB appears as red, straight, or slightly curved rods, occurring singly or in small groups, while the rest of the background, mucoid and pus cells are stained blue in colour.
The method was initially developed by Paul Ehrlich and later modified by afterwards the Franz Ziehl and Friedrich Neelsen after whom the method is named.
Video fileResources
- Laboratory Diagnosis by Sputum Smear Microscopy - The Handbook, GLI, 2013.
- Module for Laboratory Technicians, CTD, 2005.
Assessment
Question Answer 1 Answer 2 Answer 3 Answer 4 Correct answer Correct explanation Page id Part of Pre-test Part of Post-test In ZN microcopy what is the stain used Auramine Carbol fucshin Methylene blue Potassium Permanganate 2 The stain used in ZN microscopy is Carbol Fucshin responsible for the reddish color of the bacteria. Yes Yes Fluorescence Microscopy Using LED Microscope
Content
Fluorescence Microscopy is a newer and better type of microscopy where the TB bacteria are stained using a fluorescent dye using the property of Acid Fastness. The dye will fluoresce when illuminated by UV light. When the UV light source is an LED Lamp, it is called LED fluorescent microscopy.
Here the bacilli appear as slender bright yellow fluorescent rods, standing out clearly against a dark background, as can be seen in the figure below.
Figure: AFB as seen under an LED Fluorescence Microscope:
Principle of Fluorescence Microscopy
- Cell walls of Acid-fast Bacilli (AFB) is made up of Mycolic Acid. The mycolic acid creates a waxy layer, making the cell wall impermeable to acids and alkalis.

- The primary stain, a fluorescent dye called Auramine-O, binds to the cell wall of the bacilli.

- Intense decolorization by acid alcohol does not release the primary stain. Thus, the AFB retain the colour of the primary stain, while other bacteria lose the stain
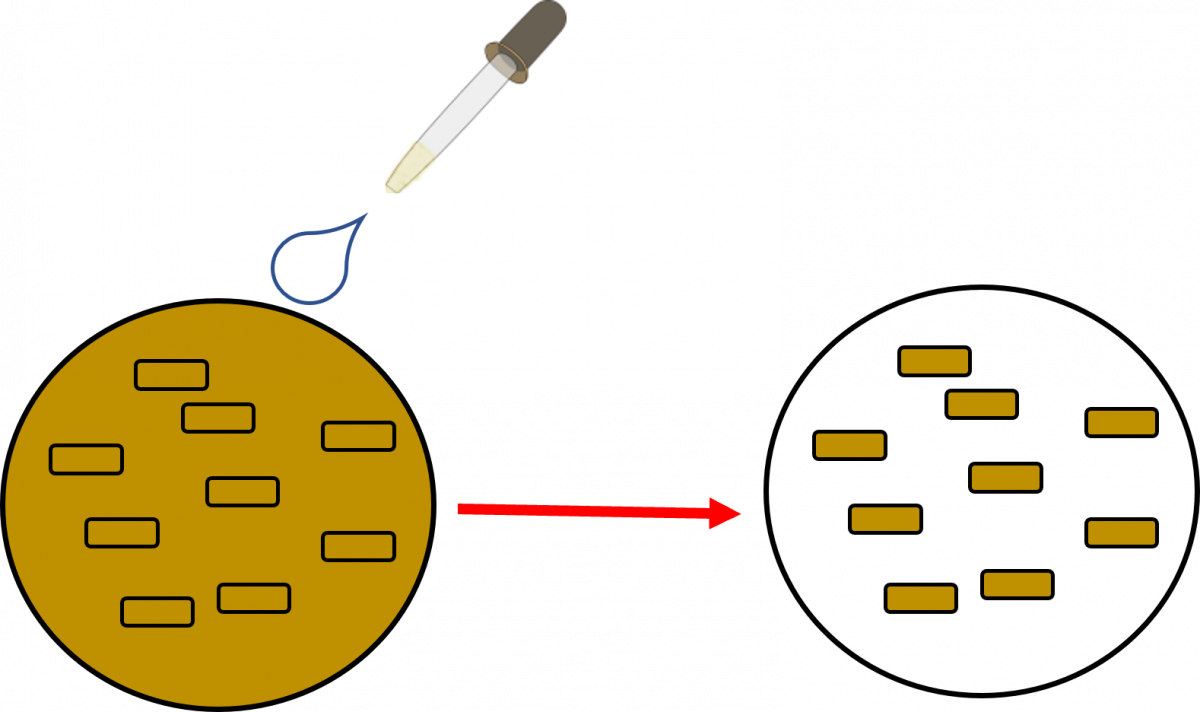
- The counterstain, Potassium Permanganate provides a contrasting background and is useful to quench background fluorescence.
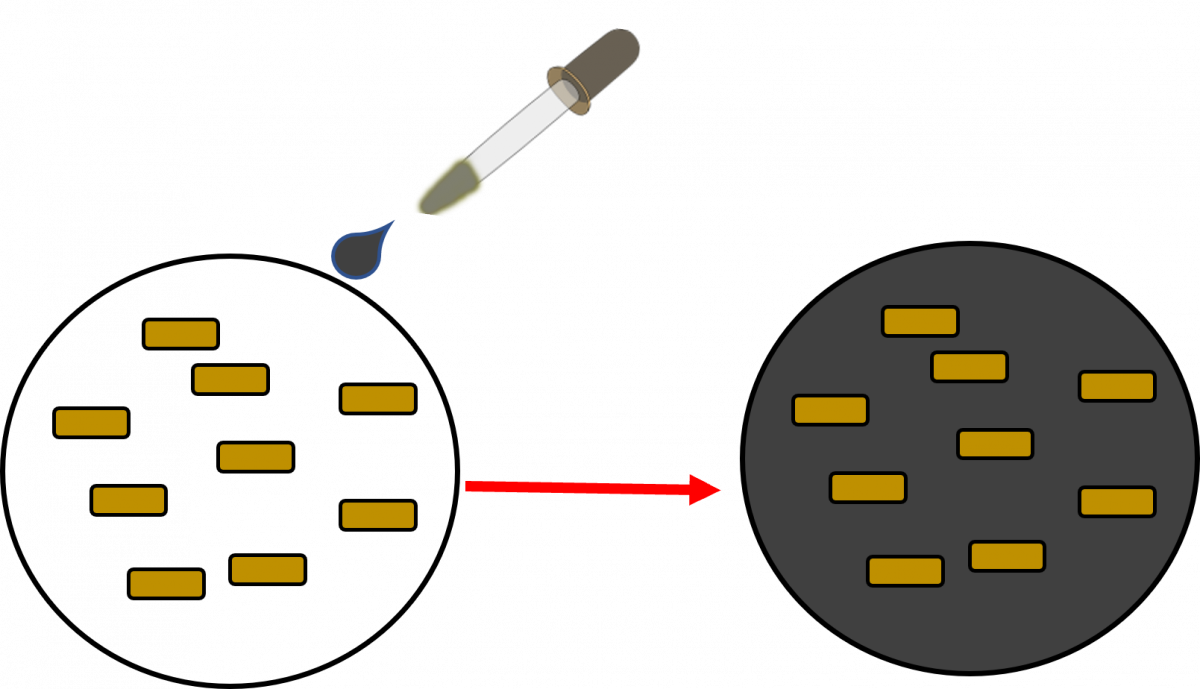
Advantages of LED-FM:
- Fluorescence LED microscopy is more sensitive (10%) than conventional ZN microscopy.
- It may be placed in existing DMCs and does not require any additional infrastructure.
- Examination of fluorochrome-stained smears takes less time.
Video fileResources
- Cell walls of Acid-fast Bacilli (AFB) is made up of Mycolic Acid. The mycolic acid creates a waxy layer, making the cell wall impermeable to acids and alkalis.
Cartridge Based Nucleic Acid Amplification Test [CBNAAT]
ContentCartridge Based Nucleic Acid Amplification Test (CBNAAT) is a rapid molecular diagnostic test. It is used for diagnosis of Tuberculosis (TB) and Rif-resistant Tuberculosis (RR-TB) in NTEP. Results are obtained from unprocessed sputum samples in about 2hours which helps in early detection and treatment of TB patients.
India has vast number of CBNAAT laboratories which are utilized for TB/RR-TB detection and Universal Drug Susceptibility Testing (UDST) under the National TB Elimination Program (NTEP).

Figure: CBNAAT Cartridge and Machine in Use (Image courtesy: USAID supported Challenge TB Project)
The CB-NAAT system detects DNA sequences specific for Mycobacterium tuberculosis complex and rifampicin resistance by Polymerase Chain Reaction (PCR). It concentrates Mycobacterium tuberculosis bacilli from sputum samples, isolates genomic material from the captured bacteria by sonication and subsequently amplifies the genomic DNA by PCR. The process identifies clinically relevant rifampicin resistance-inducing mutations in the RNA polymerase beta (rpoB) gene in the Mycobacterium tuberculosis genome in a real-time format using fluorescent probes called molecular beacons.
Video fileVideo: Cartridge-Based Nucleic Acid Amplification Test [CBNAAT] - GeneXpert Technology
Resources
- Training Module (1-4) for Program Managers and Medical Officers, NTEP, MoHFW, 2020.
- India TB Report 2021, National TB Elimination Program (NTEP), MoHFW, 2021.
Assessment Questions
Question
Answer 1
Answer 2
Answer3
Answer 4
Correct Answer
Correct explanation
Part of pre-test
Part of post-test
Under NTEP, CBNAAT is offered upfront for which of these categories?
PLHIV
Paediatric presumptive TB
Presumptive DR-TB
All of the above
4
Under NTEP, CBNAAT is recommended upfront for People living with HIV, Paediatric Presumptive TB patients, Presumptive DR-TB patients and patients notified from the Private sector.
Yes
Yes
CBNAAT requires the processing of sputum samples before testing
True
False
2
Results are obtained from unprocessed sputum samples in about 2hours from a CBNAAT machine
Yes
Yes
Truenat
ContentTruenat is an indigenous rapid molecular test platform that is currently under use in NTEP for diagnosis of TB and Rif Resistance. It is a platform utilising real-time Polymerase Chain Reaction (PCR) technology built into micro-PCR chips.
Testing on Truenat involves three components:
- Workstation (consisting of 2 devices)
- Trueprep AUTO Universal Cartridge-based Sample Prep Device for the automated extraction and purification of DNA
- Truelab Real-time micro PCR Analyzer for performing real-time PCR. It is available as 1 (Uno), 2 (Duo) or 4 (Quattro) chip ports.
- Cartridge and Chip
- Reagent kits (Sample Pre-treatment and Prep kits)

Figure: Truenat Source: MolBio Products.
Test results for MTB detection and Rif Resistance has a turn around time of 1-2 hours. Depending on the micro-PCR chips used various tests can be performed using Truenat. Truenat MTB micro-PCR chips detect Mycobacterium tuberculosis bacteria for TB diagnosis. Truenat MTB RIF micro-PCR chip is used as a reflex test to detect resistance to Rifampicin (RIF), the first-line drug for TB treatment
Truenat has many advantages. Truenat is designed to be mobile and is battery operated (~8 hours on full charge). It can be deployed in peripheral laboratories and microscopy centres with minimal or no added facilities and hence it is more point-of-care. Biosafety requirements are similar to smear microscopy. However, it is multi staged and partially automated, requiring the presence of a Lab Technician through out the test.
Resources
- Truenat MTB Kit Insert.
- Trueprep AUTO Universal Cartridge-based Sample Prep Device.
- Practical Guide to Implementation of Truenat Tests for the Detection of TB and Rifampicin-resistance, 2021.
Assessment
Question
Answer 1
Answer 2
Answer 3
Answer 4
Correct answer
Correct explanation
Page id
Part of Pre-test
Part of Post-test
Truenat is used in NTEP for: MTB detection Rif Resistance Detection INH resistance Detection MTB and Rif Resistance Detection 4 Truenat is used for MTB and Rif Resistance detection in NTEP Yes Yes The Truelab Analyzer is available in how many chip ports?
2 (Duo)
1 (Uno), 2 (Duo) and 4 (Quattro)
1 (Uno)
4 (Quattro)
2
The Truelab Analyzer is available as 1 (Uno), 2 (Duo) and 4 (Quattro) chip ports.
Yes Yes - Workstation (consisting of 2 devices)
Line Probe Assay [LPA]
ContentLine Probe Assay (LPA) is a rapid molecular test available at centralised laboratories.
The assay is based on Polymerase Chain Reaction (PCR) that can simultaneously detect Mycobacterium tuberculosis complex as well as drug sensitivity to anti-TB drugs.

Figure 1: The GenoType MTBDRplus Molecular LPA Procedure; Source: Molecular Detection of Drug-resistant Tuberculosis by Line Probe Assay.
Advantages of LPA
- Rapid molecular test. (Turnaround time: 3-5 days)
- Highly sensitive and specific.
- Performed directly from sputum smear-positive specimens and on isolates of M. tuberculosis complex grown from smear-negative and smear-positive specimens.
- Detects multiple gene mutations in anti-TB drugs.
- First-line LPA detects mutations to rifampicin and isoniazid
- Second-line LPA detects mutations to fluoroquinolones and aminoglycosides.
- Suitable for low and high-throughput labs.
Disadvantages of LPA
- Cannot be used as a point-of-care test.
- Requires appropriate laboratory infrastructure, equipment and biosafety precautions.
- Different rooms (DNA extraction, pre-amplification, amplification, post-amplification/ hybridization) are required to perform different steps (Figure 2).
- Requires trained manpower to perform tests and interpret test results.
- Stringent internal quality control is required to prevent contamination.

Figure 2: Amplification (A) and Post-amplification Laboratory (B) for LPA; Source: Molecular Detection of Drug-resistant Tuberculosis by Line Probe Assay.
Resources
- Guidelines for PMDT in India, 2021.
- Molecular Detection of Drug-resistant Tuberculosis by Line Probe Assay.
Assessment
Question Answer 1 Answer 2 Answer 3 Answer 4 Correct answer Correct explanation Page id Part of Pre-test Part of Post-test LPA can be used as a point-of-care test. True False 2 LPA cannot be used as a point-of-care test. Yes Yes Solid and Liquid Culture in TB
ContentCulturing TB Bacilli is well known and historic method for detection/ confirmation of Tuberculosis. It is a highly sensitive and specific phenotypic test; it can detect even a few viable bacilli in the sample (Upto 10 Colony Forming Units- CFUs). TB bacilli multiply in the culture and form colonies of TB bacilli which can are easily be identified.
Based on the growth media used Culture is divided in to two types, Solid and Liquid Culture methods. Types Culture:
- Solid Culture on Lowenstein Jensen media : Historic gold standard culture test. Results take usually upto 2 months (60 days).
- Modern Liquid culture systems: (e.g. BACTEC MGIT 960, BacT Alert or Versatrek etc.) Results take usually up to 42 days.
Uses
- Solid culture is the gold standard diagnostic test for TB. But it is not used for the purpose of TB diagnosis due to the long turn around time of 2 months. It is largely used for research purposes where it is used as the baseline test on which the sensitivity and specificity of other tests are calculated.
- Liquid Culture is being used for follow-up monitoring of patients on drug resistant TB treatment to detect treatment failure. Liquid culture is also used for long term follow up patients who have successfully completed treatment to detect recurrence.
- Liquid culture is used as a previous step to grow bacilli and obtain isolates prior to Drug Susceptibility Testing.
- Liquid cultures are also used in TB prevalence surveys for its high sensitivity and specificity
Resources
Kindly provide your valuable feedback on the page to the link provided HERE
Culture Drug Susceptibility Testing [CDST]
ContentCulture Drug Susceptibility Testing (CDST) is a growth-based phenotypic method used to check the susceptibility of Mycobacterium tuberculosis strains to various first and second line anti-TB drugs. Mycobacterial resistance to a particular drug is identified if there is growth observed in culture in presence of that drug.
In NTEP CDST is the standard method to detect resistance in samples of patients who have tested positive on followup. While CDST is possible on both Solid and Liquid culture, currently, the NTEP utilizes only liquid culture as a method for DST, due to faster Turn around times.
CDST testing services are available under NTEP in designated, specialized laboratories called CDST Labs both in public and private sector. Currently there are 80 such laboratories (60 certified for First Line and 49 for Second line drugs). Such designated laboratories are subject to regular external quality assessment, often by the National Reference Laboratory at that region.
Quality assured DST to R, H, Z, Mfx, Lfx, Lzd, Am, Km and Cm are available across the country.
Resources
Monitoring of Treatment by Smear and Culture
ContentSmear microscopy and culture play an important role in monitoring the response to TB treatment. In NTEP, smear microscopy and culture has been prescribed to monitor the treatment response and at specific time points based on different treatment regimens. The programme also explains the actions that need to be taken based on the follow-up results.
Monitoring of tuberculosis treatment is shown in the table below.
Table: Monitoring of TB treatment by smear and culture
TYPE OF TREATMENT
MONITORING BY SMEAR
MONITORING BY CULTURE
ACTION TO BE TAKEN FOR FOLLOW UP
Drug Sensitive Pulmonary TB
One sputum smear examination at time of completion of the intensive phase (IP) of treatment and at the end of treatment
-
If sputum is positive anytime during treatment, then drug susceptibility testing (DST) is to be done. Further treatment is guided by the DST pattern.
H mono/poly DR-TB
Monthly from month 3 onwards till the end of treatment. Conduct smear microscopy within 7 days, if the smear at month 4 or later is positive to rapidly ascertain bacteriological conversion/reversion
At the end of month 3, end of treatment (month 6 and/or 9 if applicable)
If the culture results of month 3 are positive, the culture is repeated to rapidly ascertain bacteriological conversion/ reversion. If the repeat specimen is culture negative, culture is done at the end of treatment
Shorter oral Bedaquiline-containing MDR/RR-TB regimen
Monthly from 3rd month onwards till end of IP monthly in extended IP only if previous month is smear positive ( S+ve). Conduct SM within 7 days, if the smear at 6 months is positive to rapidly ascertain bacteriological conversion/reversion.
At the end of month 3, end of month 6 and/or end of treatment.
If the culture results of month 6 is positive, collect one repeat sample immediately to rapidly ascertain the bacteriological conversion/reversion. If the repeat sample is culture negative, then conduct an end of treatment specimen collection.
Longer Oral M/XDR-TB Regimen
Examine sputum smear within 7 days and every time C&DST is done.
Monthly from month 3 onwards to end of 6 months or 7 or 8 if the previous month’s culture is positive; Quarterly month 6 or 7 or 8 onwards based on previous month’s culture results
If the smear/culture results of month 6 or any of the quarterly culture is positive, repeat culture is done to ascertain bacteriological conversion/reversion and if the repeat specimen is culture negative, then culture is done the subsequent quarter or end of treatment
Resource
Kindly provide your valuable feedback on the page to the link provided HERE
Culture Conversion
ContentSputum culture conversion - The transition in sputum culture results from a positive sample growing Mycobacterium tuberculosis to two consecutive negative cultures separated by at least 30 days.
Time to culture conversion is the interval between treatment initiation to date of specimen collection of the first (of the two) negative culture.
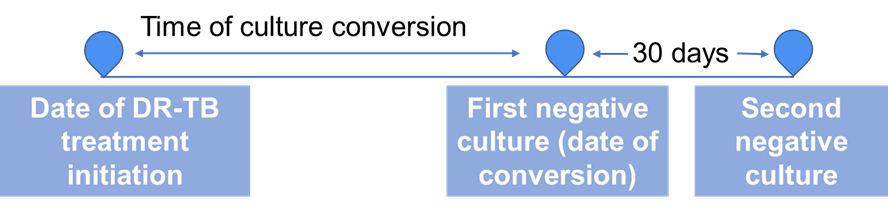
Culture conversion is important indicator to assess the effectiveness of a treatment regimen for Drug-resistant Tuberculosis (DR-TB).
All DR-TB cases are followed up periodically for culture after initiation of the treatment. Based on the culture results and other parameters, the course of treatment is decided.
Resources
- Time to Culture Conversion and Regimen Composition in Multidrug-resistant Tuberculosis Treatment; Tierney D. Harvard Library
- Guidelines for Programmatic Management of Drug-resistant Tuberculosis in India, 2021.
Kindly provide your valuable feedback on the page to the link provided HERE
-
CDST_LT: Diagnostic network and hierarchy in NTEP
FullscreenLaboratory Hierarchy and Network
ContentNTEP laboratory network is comprising of National Reference Laboratories (NRLs), state level Intermediate reference laboratories (IRLs), Culture & Drug Susceptibility Testing (C & DST) laboratories and peripheral level laboratories. Peripheral level laboratories consist of designated microscopy centres (DMCs) and NAAT labs.
NTEP has a quality assured laboratory network for bacteriological examination of sputum in a 3-tiered system.
Figure: Laboratory network of NTEP
Resources:
Kindly provide your valuable feedback on the page to the link provided HERE
National Reference Laboratories [NRL]
ContentThe National Reference Laboratories (NRLs) constitute the third tier of the National Tuberculosis Elimination Programme (NTEP) laboratory network hierarchy.
They provide quality assurance and certification services for the Culture and Drug Susceptibility Testing (C&DST) labs and coordinate with the World Health Organisation (WHO) Supranational Reference Laboratory (SNRL) network.
There are six designated NRLs which are delineated in the figure below.
Image
Figure: Six National Reference Laboratories under NTEP in India
NIRT, Chennai, in addition to being one of the NRLs is also one of the WHO designated SNRLs for the Southeast Asia Region.
Resources
- Guidelines for Programmatic Management of Drug-resistant TB in India, 2021.
- NTEP Laboratory Network: Overview.
Question
Answer 1
Answer 2
Answer3
Answer 4
Correct Answer
Correct explanation
Part of pre-test
Part of post-test
How many designated NRLs are there in India?
8
6
4
2
2
There are 6 designated NRLs in India.
Yes
Yes
Who is responsible for quality assurance and certification services for the C&DST labs?
IRLs
State TB Cell
NRLs
Central TB Division
3
NRLs provide quality assurance and certification services for the Culture and Drug Susceptibility Testing (C&DST) labs and coordinate with the World Health Organisation (WHO) Supranational Reference Laboratory (SNRL) network.
Yes
Yes
Roles of NRLs
Content- National Reference Laboratories (NRLs) conducts annual on-site evaluation/supervisory visits to laboratories for assessing the quality of microscopy, culture and drug susceptibility test (C&DST), and for improvement of the overall laboratory quality.
- NRLs also assist Central TB Division (CTD), in developing laboratory guidelines, standard operating procedures (SOPs), and conduct training to state-level Intermediate reference laboratories (IRLs) and other technical issues.
- NRLs conduct C&DST training to the IRLs, and develop SOPs for the technical procedures, equipment maintenance, infection control and recording and reporting.
- NRLs are also responsible for offering second-line drug susceptibility tests (DST) for multi-drug resistant TB (MDR-TB) treatment failures.
- NRLs are responsible for the accreditation of the mycobacteriology laboratory for culture and drug sensitivity testing under the National Tuberculosis Elimination Program (NTEP).
- In addition, NRLs are also responsible for the conduct of research for the programme and evaluation of newer tools for the diagnosis of TB.
- The National Institute for Research in Tuberculosis (NIRT) Chennai, the Supranational Reference Laboratory (SRL) of the region is responsible for the external quality assurance of the other 5 NRLs. NIRT is in turn quality-assured through the SRL coordinating laboratory at Antwerp, Belgium.
Assessment Questions
Question
Answer 1
Answer 2
Answer3
Answer 4
Correct Answer
Correct explanation
Part of pre-test
Part of post-test
What are the functions of National Reference Laboratories?
Providing Culture and DST training to the IRLs
Developing SOPs for the technical procedures
Offering second-line DST
All of the above
4
All the functions stated are performed by the National Reference Laboratories.
Yes
Yes
Which institute is responsible for the external quality assurance of NRLs?
SRL
CTD
National Institute for Research in Tuberculosis, Chennai
National Tuberculosis Institute, Bangalore
3
The National Institute for Research in Tuberculosis (NIRT) Chennai, the Supranational Reference Laboratory (SRL) of the region is responsible for the external quality assurance of the other 5 NRLs. NIRT is in turn quality-assured through the SRL coordinating laboratory at Antwerp, Belgium.
Yes
Yes
Intermediate Reference Laboratories [IRL] and their role
ContentSome Culture and Drug Susceptibility Test (C&DST) laboratories host an Intermediate Reference Laboratory (IRL) under the National TB Elimination Programme (NTEP).
There is at least one IRL per state at an identified location, usually in a secondary or tertiary level public health facility. There are 34 IRLs in India.
The IRLs are responsible for:
- Undertaking training on laboratory technologies for district and field level staffs
- Conducting on-site evaluation visits to districts for sputum microscopy at least once a year
- Undertaking panel testing of Senior TB Laboratory Supervisors (STLS) at each district linked to it
- Ensuring the proficiency of staff performing National Tuberculosis Elimination Programme (NTEP) smear microscopy activities by providing training to laboratory technicians and STLS
Resources
- Guidelines for Programmatic Management of Drug-resistant TB in India, 2021.
- NTEP Laboratory Network: Overview.
Kindly provide your valuable feedback on the page to the link provided HERE
CDST labs and their role
ContentUnder the National Tuberculosis Elimination Programme (NTEP), many labs are established at the regional level within states for providing Culture and Drug Susceptibility Testing (C&DST) facilities for presumptive TB/DRTB and for TB/DRTB patients.
- C&DST laboratories are mostly located in intermediate reference laboratories (IRLs) or medical colleges.
- There are 42 C&DST laboratories established under the programme in different geographies.
- Dedicated human resources are provided for the laboratories under the programme.
- Districts are linked with laboratories for providing facilities for Culture and DST using:
- Phenotypic Methods (Solid – Lowenstein Jensen (LJ), and Liquid Culture – Mycobacteria Growth Indicator Tube (MGIT))
- Genotypic technology (Line Probe Assay (LPA) and Cartridge Based Nucleic Acid Amplification Test (CBNAAT))

Figure: Culture and Drug Susceptibility Testing (C&DST) facility,
Source: The Foundation For Innovative New Diagnostics (FIND)Resources
- Guidelines for Programmatic Management of Drug Resistant TB in India, 2021
- Training Modules (1-4) for Programme Managers and Medical Officers; New Delhi, India: Central TB Division, July 2020
Kindly provide your valuable feedback on the page to the link provided HERE
NAAT Labs and their role
ContentThe National Tuberculosis (TB) Elimination Program (NTEP) has a network of Nucleic Acid Amplification Tests (NAAT) laboratories coupled with Designated Microscopy Centers (DMCs) to form the backbone of the diagnostic component of TB services.
Nucleic Acid Amplification Tests (NAAT) laboratories includes Cartridge-based NAAT (CBNAAT) and TrueNat tests. These tests detect tuberculosis as well as rifampicin resistance and are more sensitive than smear microscopy.
Functions of Nucleic Acid Amplification Test (NAAT) Laboratories:
- Acting as a hub for collection of samples from public and private health facilities (spokes)
- Universal Drug Susceptibility Testing (UDST) to rule out rifampicin resistance among confirmed TB patients
- Timely provision of NAAT test result to the TB patient, medical officer of the concerned health facility and NTEP staff for related actions
- Acting as a sample dispatch center for the Culture DST laboratory for subsequent processing of samples for first-line line probe assay (LPA) and second-line drug resistance testing utilizing second line LPA and liquid culture DST
- Recording and reporting including digitization of diagnostic process from collection to test result in NTEP Nikshay portal and Laboratory Information Management System
- Management of supplies and logistic associated with laboratory logistic (CBNAAT cartridges and TrueNAT chips) and reporting any additional requirement thereof
- Supporting the quality assurance activities undertaken by District or Intermediate Reference Laboratory under NTEP
- Support health system in carrying out special drives for vulnerable and at-risk population and their testing directly by CBNAAT (slum population, diabetic population, smoker, malnourished people, patients of silicosis and kidney dialysis etc.)
Resources
- RNTCP Technical and Operational Guidelines for TB Control in India, 2016.
- Guidelines for Programmatic Management of Drug Resistant Tuberculosis in India, 2021.
Kindly provide your valuable feedback on the page to the link provided HERE
Designated Microscopy Centre [DMC]
ContentSputum microscopy diagnostic services under the National TB Elimination Programme (NTEP) are provided by the Designated Microscopy Centres (DMCs) established at the Peripheral Health Institution (PHI) level, where a functional binocular microscope and a trained Laboratory Technician (LT) is available. Light Emission Diode Fluorescent microscopes are provided to high-load PHI-DMCs such as that of the medical colleges.
Based on latest directives, a DMCs may be established at all PHIs (Public and Private) of the country as needed. It is mandatory to have a DMC at all medical colleges in the country.
As molecular technologies like Truenat are also used in DMCs, NTEP has planned to rename DMCs as TB Diagnosis Centres (TDCs).
Criteria to be a DMC
The DMCs should satisfy the following criteria:
- NTEP-trained Laboratory Technician (LT) should be present.
- A functional binocular microscope should be present in the laboratory.
- Physical infrastructure in the laboratory should meet NTEP guidelines.
- Daily new adult outpatient cases of at least 60-100 and/or workload of at least 3-5 sputum smears per day for the LT in the laboratory.
DMCs in the public sector, at the onset of the programme, are provided with funds to undertake minor civil works to build up their physical infrastructure and are provided with binocular microscopes.
Human Resources Norms
- For the purpose of NTEP, a PHI is a health facility which is manned by at least a Medical Officer (MO).
- In addition to the MO and LT, there is 1 TB Health Visitor (TBHV) per one lakh urban population to support the urban TB control activities.
Other Criteria
Microscopy Centres may be established beyond population norms in medical colleges, corporate hospitals, Employee State Insurance Corporation (ESIC), railways, Non-government organisations (NGOs), private hospitals, Ayushman Bharat - Health and Wellness Centres (AB-HWCs), etc.
Before designating a DMC in other sectors, there should be a formal agreement by the hospital/ laboratory to take part in the External Quality Assurance (EQA) and to allow the concerned NTEP staff to supervise as per the NTEP guidelines.
If the above criteria are met by any private laboratory, the lab is considered for establishing a DMC.
- To provide better access for diagnosis of TB, all PHIs, wherever LTs and binocular microscopes are available, can be upgraded to a DMC irrespective of the population norms or OPD attendance.
- All DMCs should comply with the Quality Assurance (QA) mechanisms as per the EQA guidelines.
Resources
- NTEP Training Modules 1-4 for Programme Managers & Medical Officers, 2020.
- Operational Guidelines for TB Services at Ayushman Bharat Health and Wellness Centres.
Assessment Questions
Question
Answer 1
Answer 2
Answer3
Answer 4
Correct Answer
Correct explanation
Part of pre-test
Part of post-test
The DMC is an NTEP diagnostic facility at the PHI level
TRUE
FALSE
1
The DMC is an NTEP diagnostic facility at the PHI level
Yes
Yes
DMCs are established only in a public sector facility
True
False
2
If a lab/facility meets the criteria of DMC, the facility is considered for establishing a DMC
Yes
Yes
Kindly provide your valuable feedback on the page to the link provided HERE
Functions of a Designated Microscopy Centres [DMC]
ContentFunctions and Integrated Services of the DMC
- Testing of Sputum samples by Microscopy.
- Request/ referral for microscopy or Nucleic Acid Amplification Test (NAAT) or Culture and Drug Susceptibility Test (C&DST) or Chest X-ray (CXR) or Tuberculin Skin Test (TST) is generated at the PHI-DMC, as well as follow-up tests.
- Maintain consumables and logistics required for testing/ packaging and transport.
- Maintain TB laboratory registers for recording and reporting.
- Notify every TB patient in Nikshay at the earliest and update information of patients on comorbidity, treatment adherence, treatment outcome, contact investigation and TB Preventive Treatment (TPT).
- Biomedical waste management for the waste generated at DMCs.
- A DMC is required to participate in the External Quality Assurance system(EQA) of NTEP to ensure standardized quality diagnostic testing.
Resources
Kindly provide your valuable feedback on the page to the link provided HERE
Sputum Collection centres
ContentTo increase access to diagnostic services, NTEP has a provision for sputum collection centres in areas where the health facility is not equipped with key requirements to conduct sputum microscopy, molecular tests, drug susceptibility testing or follow up examinations.
Sputum collection centres are dedicated locations where sputum samples are collected, packaged and then transported to nearby TB diagnostic centres. It could be attached to any near-by health-facility as well.
Requirements of a Sputum Collection Centre
To function as sputum collection centres, the following is essential:
- Linkage/ mapping (time and distance) to testing laboratory
- Availability of adequate number of sputum cups and falcon tubes, logistics for sample packaging and transport
- Identification of open areas for sputum collection
- Staff trained in NTEP guidelines on sputum collection, sample packaging and transport, complete and correct documentation of laboratory request form, and infection control practices
- Feasibility and financial measures required for sample transport
- Inclusion of local volunteers, courier services, sample transportation under National Health Mission Free Diagnostic Services or other mechanisms as decided by the state/district
- Availability of Information, Education and Communication (IEC) material, training modules, and job-aids
Sputum collection centres are established in:
- Ayushman Bharat Health and Wellness Centres/Sub-centres
- Urban primary health centres
- Tribal, hilly, desert and difficult-to-reach areas of the country
Resources
- Training Modules for Programme Managers and Medical Officers
- Operational Guidelines for TB Services at Ayushman Bharat Health and Wellness Centres
- Mycobacteriology Laboratory Manual, GLI Initiative, 2014
Assessment:
Question Answer 1 Answer 2 Answer 3 Answer 4 Correct answer Correct explanation Page id Part of Pre-test Part of Post-test Under NTEP, where are sputum collection centres established to increase access to diagnostic services? Tribal areas Ayushman Bharat health and wellness centres Difficult-to-reach areas All of the above 4 To increase access to diagnostic services, sputum collection centres are established in Ayushman Bharat health and wellness centres, Urban health centres, tribal, hilly, desert and difficult-to-reach areas of the country. Yes Yes
-
CDST_LT: TB diagnosis
FullscreenPresumptive TB
ContentPresumptive TB case refers to a patient who presents with symptoms or signs suggestive of TB disease (previously known as a TB suspect) and where further diagnostic workup including bacteriological investigation is required.
Presumptive TB can be categorized into
-
Presumptive Pulmonary TB (P TB) - Symptoms are directly related to lungs (Cough, hemoptysis)
-
Presumptive Extra Pulmonary TB (EP TB) - Symptoms/ signs are specific to an extra pulmonary site (example: Lymph node swelling)
-
Presumptive Pediatric TB - Symptoms of TB in young children are more difficult to identify and can be more general (fever, weight loss)
Resources:
- Technical and Operational Guidelines for TB Control in India 2016
- Definitions and reporting framework for tuberculosis
Kindly provide your valuable feedback on the page to the link provided HERE
-
Principles of TB Diagnosis under NTEP
ContentThe National TB Elimination Program (NTEP), promotes the following principles to diagnose TB:
- Try to establish the microbiological confirmation for all cases
- Use rapid molecular diagnostics upfront wherever possible for diagnosis of TB and early identification of resistance to treating drugs.
- Focus more on quality sample collection and timely transportation for a better microbiological confirmation
Microbiological Confirmation for All Cases
Microbiologically confirmed TB refers to a presumptive TB case from whom a biological specimen is positive for acid fast bacilli smear microscopy, or positive for Mycobacterium tuberculosis on culture, or positive for TB through Rapid Diagnostic molecular tests - Nucleic Acid Amplification Test (NAAT) and Line Probe Assay (LPA). Establishing microbiological confirmation is key for all TB cases. Clinically diagnosing TB should be limited only to very few patients where, in-spite of high suspicion, microbiological confirmation could not be established, even after all possible efforts. The entire diagnostic algorithm puts utmost efforts to establish the microbiological evidence in a case of TB.
Upfront Rapid Molecular Diagnostics
Knowing the drug resistance pattern at the earliest is key for success of the treatment. Hence, the current policy highlights the importance of using molecular diagnostic test upfront wherever possible.
Complete diagnosis of TB is achieved by:
- Offering NAAT (CBNAAT/ Truenat) to all notified new patients and to test for resistance to Rifampicin. This is termed as Universal Drug Sensitivity Test (DST) for Rifampicin. Efforts are being made to collect specimen from all TB patients for NAAT at baseline.
- Testing individuals belonging to key population groups (clinically, socially vulnerable), those with extra pulmonary TB, people living with HIV and paediatric patients (after X-ray screening). They are directly referred for TB testing by NAAT
- For upfront NAAT, one specimen is tested using NAAT and if TB is detected, the other sample is used for further cascade testing by LPA and liquid culture
Quality Sample Collection and Transport
For TB diagnosis, it is essential that a good sputum sample is collected. A good specimen consists of recently discharged material from the bronchial tree with minimum amount of oral or nasopharyngeal material, presence of mucoid or mucopurulent material and should be 2-5 ml in volume. The specimen is collected in a sterile container after rinsing of the oral cavity with clean water. The collected specimens should be packaged and transported to the laboratory as soon as possible after collection.
Resources
- Guidelines for Programmatic Management of Tuberculosis Preventive Treatment, Central TB Division, MoHFW 2021
- Training Modules for Programme Managers and Medical Officers,Central TB Division, MoHFW 2020
- Guidelines on Airborne Infection Control, Directorate General of Health Services, MoHFW 2010
Assessment
Question Answer 1 Answer 2 Answer 3 Answer 4 Correct answer Correct explanation Page id Part of Pre-test Part of Post-test What are the key principles of TB diagnosis under NTEP?
Upfront testing for vulnerable groups Microbiological confirmation of all cases Good sample collection and transport All of the above
4 The key principles of TB diagnosis under NTEP are: microbiological confirmation for all cases, use upfront NAAT, quality sample collection and transport, and practicing universal precautions and AIC measures.
Biological Specimen for Diagnosis of TB
ContentFor laboratory diagnosis of TB, different biological specimens are used.
Pulmonary TB: Sputum sample is used. Sputum is a thick fluid produced in the lungs and in the adjacent airways. Normally, a spot sample and a fresh morning sample is preferred for the bacteriological examination of sputum.
Extra Pulmonary TB:
Resources:
Kindly provide your valuable feedback on the page to the link provided HERE
General process flow for testing in NTEP
ContentThe process for testing is initiated with a request for test and ends with the reporting of test results. In built into the process of testing is also the process of specimen collection and transportation.
1. Request for a test: This is the first step initiated when the requirement for a test is identified. This event is at the time of identification of presumptive TB/ DRTB, follow-up point (end of IP/CP, post treatment follow-up). The request may be initiated by the medical officer (MO) or the health staff at the Peripheral Health Institute (PHI) citing the reason for testing and the type of test required. The request is directed towards a laboratory where the required test is available. The request for test can be performed in Nikshay for any case that is already enrolled with an existing Patient ID. Requesting for test in Nikshay is analogous to filling up the physical request form Annexure 15A and generates a Test ID/ Test Request ID.
2. Patient Referral/ Sample collection and transportation: Following the request for test, the next step is to physically refer the patient to the corresponding laboratory, or collecting the appropriate biological sample and initiating its transportation to the lab. If biological sample is collected, the details of the sample need to be added under the request for test in Nikshay and the sample needs to be appropriately labelled and the corresponding details of the request test attached.
3. Performing the test: Once the sample has been received successfully at the destination laboratory, the lab technician (LT) updates the sample/ test record in Nikshay and initiates the relevant protocol for testing and follows through till results availability.
4. Reporting results: Once the results are available it needs to be updated against the corresponding request for a test and it is visible to all relevant stakeholders in Nikshay. If only Annexure 15A is available, the results need to be updated there and needs to be manually communicated to the personnel initiating the request for test.
Resources
- Training Modules for Programme Managers and Medical Officers
- Guidelines for Programmatic Management of Tuberculosis Preventive Treatment
Assessment
Question
Answer 1
Answer 2
Answer 3
Answer 4
Correct answer
Correct explanation
Page id
Part of Pre-test
Part of Post-test
Which of the following statements are correct?
Under NTEP, the process for testing is initiated with a request for test on only Nikshay.
Test requests for presumptive DR-TB cases are initiated only by specialists.
Test requests for all presumptive cases are initiated by the medical officer and/or other health staff at the Peripheral Health Institute (PHI).
All of the above
3
Test requests for all presumptive cases are initiated by the medical officer and/or other health staff at the Peripheral Health Institute (PHI).
Yes Yes Requesting a Test on Nikshay
ContentOnce a presumptive TB patient is identified, the patient is enrolled online by a healthcare worker or doctor in Nikshay online portal. For diagnosis of Tuberculosis, the treating physician can request a test utilizing the request test option of Nikshay online portal. The step-by-step approach for test request is as follows:
Step 0: Go to the Patients Page.
Step 1: Select the “Tests” tab.
Step 2: Click the “Add Test” button.
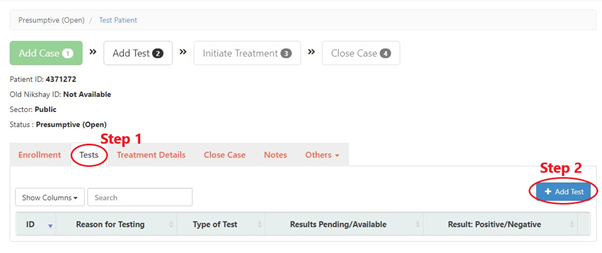
Step 3: Fill the form.
Step 4: Select the “Test Status” as “Results Pending”.
Step 5: Click the “Add Test” button by selecting the appropriate test for the patient.
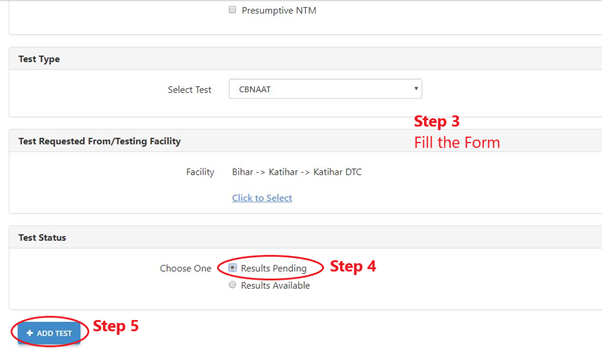
In a situation where the patient is referred to another health facility for TB testing, one needs to select the test requested along with the facility name where the patient will visit for undergoing the TB test. the results are added by the concerned healthcare worker only after the test is conducted and the result is available.
In the absence of such results, it will show ‘Result pending’ instead of ‘Result available’ status.
Video fileVideo: Process to add tests on Ni-kshay
Resources
Kindly provide your valuable feedback on the page to the link provided HERE
-
CDST_LT: Case finding methods under NTEP
FullscreenApproaches to TB Case Finding
ContentPeople who have been exposed to patients with infectious TB are known as TB contacts; they constitute a high-risk group for TB. Case finding investigation contributes to the early detection of TB cases, and results in identifying a significant number of additional patients.
Figure: Approaches to Tuberculosis Case Finding
Active case-finding requires systematic screening and clinical evaluation of populations who are at high risk of developing TB, such as people living in slums, tribal areas, congregate settings, persons who are household contacts of TB cases
Resources:
Kindly provide your valuable feedback on the page to the link provided HERE
Passive Case Finding
ContentPassive case finding is essentially where the patient self reports to the health care provider with symptoms. This requires that affected individuals are aware of their symptoms, have access to health facilities, and are evaluated by health workers or volunteers who recognise the symptoms of TB and link those individuals for TB testing services.
This approach to case finding has the least effort and cost and is a minimum expectation. In a Peripheral Health Institution (PHI), it is estimated that about 2-3% of new adult outpatients are symptomatic that require referral for TB diagnosis (presumptive TB cases).
Passive case finding may miss TB patients if :
- The disease is mild/ transient.
- Access to healthcare is poor.
- Health providers do not have an adequate index of suspicion and are unable to reliably link respiratory symptoms to TB.
Resources
Assessment
Question Answer 1 Answer 2 Answer 3 Answer 4 Correct answer Correct explanation Page id Part of Pre-test Part of Post-test Which of the following can be considered a passive case finding? TB case finding for all patients attending an HIV clinic. TB case finding in all inmates of an elderly home. Patients attending a PHC with symptoms are referred for testing by the doctor. TB case finding among household contacts of a TB case. 3 All other examples except those attending PHC referred for TB testing are cases of active or intensified case finding effort. Yes Yes What may cause a passive case finding to miss cases? Healthcare providers fail to notify the case. Healthcare providers do not refer cases for TB testing. There are no health facilities in the area. Both 2 and 3 4 Healthcare providers failing to notify cases is missing notification and not related to passive case finding. Yes Yes Active Case Finding
ContentSystematic screening of all individuals of a defined population is known as active case finding. It is applied outside of health facilities at the community level by the health system.
Objective of ACF is to:
- identify cases early, initiate prompt treatment, reduce risk of poor treatment outcomes and reduce risk of further transmission of TB
- to provide access to diagnosis services to populations that would have been otherwise unreached
It is effort intensive and is recommended only in population groups where there is estimated high case load. In NTEP, ACF is recommended only to be performed in Key / vulnerable population.
ACF can also be clubbed with suitable ACSM campaigns to create awareness about the signs and symptoms and about TB in the target population/ community. It can also be combined with other health activities/ campaigns (such as Pulse Polio/ Leprosy screening/ population based screening for NCDs) for increased efficiency.
Resources
- Training Modules for Programme Managers and Medical Officers.
- Active TB Case Finding, Guidance Document.
- WHO recommendations for Systematic Screening for Active Tuberculosis
Assessment
Question Answer 1 Answer 2 Answer 3 Answer 4 Correct answer Correct explanation Page id Part of Pre-test Part of Post-test Which of the following is not a primary objective of ACF? Increase TB notification Early identification of cases. Reduce the risk of transmission of TB. Reduce the risk of poor treatment outcomes. 1 Notification is not a primary objective of ACF. Yes Yes Intensified Case Finding
ContentIntensified Case Finding (ICF) is a case finding approach between Active and Passive approaches. Here individuals coming in contact with the health system through any activity are screened actively for symptoms of TB and referred for testing.
This approach brings the benefit of active case finding approach by active screening for TB symptoms, but does limit the extensive effort required by restricting to only those people who has some or the other healthcare problem. This approach is considered for people attending a healthcare facility.
Some examples of ICF are screening for TB symptoms and referral for testing in:
- all cases attending an HIV clinic.
- among children with malnourishment who attend a nutrition clinic.
- all mothers attending the antenatal clinics
Resources
Assessment
Question Answer 1 Answer 2 Answer 3 Answer 4 Correct answer Correct explanation Page id Part of Pre-test Part of Post-test Which of the following is an example of an intensified case finding?
Systematic screening for TB of all contacts of TB cases. Screening all cases attending an OPD with respiratory symptoms for TB testing. Referring cases that report more than 2 weeks of cough from an OPD for TB testing. Screening all people belonging to a slum for TB symptoms. 2 Systematic screening of TB contacts and those belonging to a slum population are examples of active case finding.
Referring to cases that report TB symptoms is a passive case finding.
Yes Yes Bidirectional Screening
ContentBidirectional screening is a method to identify cases in diseases which have predisposition to each other or has a significant influence on each other. For example TB and HIV, where having HIV increases risk of developing TB and cases with TB would have poor outcomes if co-infected with HIV.
Screening for TB is done through four-symptoms complex based screening or through Chest X-ray. Screening for the linked disease is carried out as per the policies of the corresponding health program.
Bi-directional screening policies are implemented by various disease control programs. For example, with NTEP the following disease control efforts implement a bidirectional screening policy:
- HIV through NACO
- COVID19
- Diabetes Mellitus (DM) through NPCDCS
- Tobacco through National Tobacco Control Program
Both programs monitor bidirectional screening, referral and testing as per their own policies.
Resources
Assessment
Question Answer 1 Answer 2 Answer 3 Answer 4 Correct answer Correct explanation Page id Part of Pre-test Part of Post-test Bidirectional screening for TB is not done in which of the following conditions? Diabetes Tobacco abuse/ addiction Pregnancy COVID-19 3 Although pregnant mothers may be screened for TB as a part of intensified case finding, all TB cases are not actively/ routinely screened for pregnancy. Yes Yes
-
CDST_LT: Role of the senior LT and related stakeholders in TB Care
FullscreenDuties and responsibilities of the LT at the DMC
ContentThe major duties and responsibilities of the Laboratory Technician at the Designated Microscopy Centre (DMC) are to:
- Follow the standard operating procedures for sample collection, sputum smear microscopy and Nucleic Acid Amplification Test (NAAT)
- Maintain the Tuberculosis (TB) Laboratory Register and early submission of the results to the medical officer managing the patient, which should also be updated in the Nikshay online portal in real-time
- Coordinate with other staff to ensure that presumptive TB cases and symptomatic contacts of TB patients receive sputum containers with the necessary instructions to undergo sputum examination/NAAT
- Assist the medical officer of the peripheral health institution (MO-PHI) in the identification of presumptive drug-resistant TB patients and ensure the collection and transportation of sputum specimens for NAAT/culture and drug susceptibility test according to the guidelines
- Organize and supervise the disposal practices of contaminated lab material as detailed in the Laboratory Manual
- Assist the Senior Tuberculosis Laboratory Supervisor (STLS) in the implementation of the National Tuberculosis Elimination Programme (NTEP) Lab Quality Assurance
- Assist in the implementation of new TB diagnostic tools in NTEP
- Facilitate change management with respect to use of Information and Communications Technology (ICT) and Nikshay tool for concerned data entry, validation, and its use for public health actions
- Any other jobs assigned by the reporting officer
Resources
- DO letter - TOR and need norms for NTEP staff, 2021.
- Training Module (1-4) for Program Managers and Medical Officer, NTEP, MoHFW, 2020.
Kindly provide your valuable feedback on the page to the link provided HERE
Role of Medical Officer at and around a DMC in TB Diagnosis
ContentThe Medical Officer Designated Microscopy Centre (MO-DMC) at a Primary Health Facility is appointed from the General Health System. The MO-DMC is responsible for activities at DMC under the National TB Elimination Programme (NTEP).
Key Responsibilities of MO-DMC include
1. Screening and diagnosis
· Clinical examination of all TB cases should be done by the MO.
· The MO should screen/refer:
o All identified presumptive pulmonary TB cases for sputum smear microscopy, chest X-ray and presumptive Extra-pulmonary Tuberculosis (EPTB) cases for appropriate investigations
o Presumptive TB cases with a negative sputum result to be referred for chest X-ray, followed by Cartridge-based Nucleic Acid Amplification Test (CBNAAT) as per diagnostic algorithm to ensure no TB case is missed
o All diagnosed TB patients for Nucleic Acid Amplification Test (NAAT) for early diagnosis of resistance to Rifampicin (Rif)
o All Rif-sensitive TB patients for first-line LPA testing
o All presumptive TB patients for HIV testing
o All diagnosed TB patients for HIV testing
o HIV positive patients for TB by four symptom complex screening
o All HIV positive TB patients to Antiretroviral Therapy (ART) centre for initiation of ART and Co-trimoxazole Prophylaxis Therapy (CPT).
2. Treatment initiation, follow up and treatment outcome
· The MO should fill the original treatment card with details of treatment regimen according to weight-band and Drug Susceptibility Testing (DST) pattern.
· It is the responsibility of the MO to ensure that all the diagnosed smear-positive patients start treatment or are referred for treatment.
o All patients who are sensitive to Isoniazid (H) & Rifampicin (R) and all patients whose H & R status is not known should be initiated on first line anti-TB treatment.
· The MO is responsible for clinically following-up the patient once in a month to:
o Identify any ADR early
o Assess clinical improvement
o Support follow-up by laboratory investigations, whenever necessary
o Control comorbid conditions like HIV and diabetes by appropriate treatment
o Screen all patients for presence of symptoms of TB at the end of 6th, 12th, 18th and 24th month after completion of treatment and do a sputum culture in the presence of symptoms to diagnose recurrent TB.
3. Recording, reporting and TB notification
· The NTEP Request Form for examination of biological specimens should be filled up completely by the MO.
· The MO should coordinate with the STLS to ensure that tuberculosis-related laboratory services are properly performed and recorded by the laboratory technician.
· Results recorded in the laboratory register, treatment cards and the TB Notification Register should be verified and ensured that they are consistent.
· The MO should maintain TB Notification Register for patients diagnosed and transferred-in.
· The MO should ensure that the treatment details are entered in Nikshay immediately.
· Detailed description of symptoms and signs of ADRs to anti-TB drug should be recorded in TB Treatment Card by the MO.
· The treatment outcome has to be recorded on the Treatment Card, Nikshay and the TB Notification register within one month of the event. Declaration of the treatment outcome has to be decided upon and signed with date by the MO.
· The MO should ensure updating of Notification Register and Nikshay entry by the designated staff:
o If any smear-positive patients are not entered in the TB Notification Register and are on treatment
o For patients who have not been put on treatment after tracing them and putting them on treatment immediately
o After collecting the bank account details of the patient for Direct Benefit Transfer of Nikshay Poshan Yojana.
4. Monitoring and supervision
· Every week, the MO of the DMC should review the TB Laboratory Register to ensure that correct number of sputum smear examinations (two per presumptive TB case) are being performed for diagnosis.
· The MO of the DMC should cross-check the results of the sputum examination in the TB Register with that of TB Laboratory Register and the TB Treatment Card.
· The MO of the DMC should check the Tuberculosis Laboratory Register to make sure that all the columns have been completed.
o The MO of DMC is responsible for determining the amount of reagents and consumables the DMC needs every month.
o The MO should ensure uninterrupted supply of drugs; monitor monthly replenishment of stock to treatment supporter if drugs are not already
given and update in drug stock register and in Nikshay Aushadhi through designated staff.· The District TB Officer (DTO) conducts Random Blinded Rechecking (RBRC) of sputum smear microscopy and gives feedback and corrective actions to Lab technicians through MO-DMC.
Resources
Training Modules for Programme Managers and Medical Officers.
Assessment
Question
Answer 1
Answer 2
Answer 3
Answer 4
Correct answer
Correct explanation
Page id
Part of Pre-test
Part of Post-test
What are the key duties of a Medical Officer at DMC?
Screening and diagnosis
Advocacy and support to private practitioners
Treatment initiation, follow-up, treatment outcome
All the above
4
The Medical Officer (MO) at the DMC is responsible for screening, diagnosis, treatment initiation, follow-up, treatment outcome, monitoring and supervision, recording, reporting, TB notification, advocacy and support to private practitioners.
Yes
Yes
Role of Peripheral Health Care workers around a DMC in TB Care
ContentPeripheral Health Care Workers (PHWs) including Community Health Officer (CHO), Auxiliary Nurse Midwife (ANM) and Multi-purpose health worker (MPHW) are central to primary health care and service delivery. They play important an role in TB care at Peripheral Health Institutes (PHIs) and Designated Microscopy Centres (DMCs).
Their responsibilities include:
1. Vulnerable population mapping: vulnerability assessment and mapping of vulnerable population (diabetic patients, patients on immunosuppressants, alcoholics and smokers etc) in communities is done by PHWs.
2. Screening and referral for testing:
PHWs are involved in:
- screening of household/workplace contacts and other contacts of TB patients as eligible in the local context
- periodic active case finding among identified vulnerable population for TB/Latent TB Infection
- referring presumptive TB patients promptly to the nearest microscopy or molecular laboratory through laboratory request forms
- registration of referred cases in Nikshay as presumptive TB patient
- providing sputum container to persons with symptoms of TB and counselling for collection of good quality sputum in the morning
- sample packaging and transport to TB laboratories for testing
- follow-up sputum examination
CHO is responsible to ensure availability of adequate sputum collection containers (sputum cups and falcon tubes), logistics for sample packaging and transportation.
3. Treatment initiation: It is the responsibility of the PHWs in coordination with NTEP staff to organize and ensure treatment initiation for the patient. They decide upon a convenient location for drug administration, identification of treatment supporter and supply of drugs to treatment supporter
4. Coordinating treatment support:
- PHWs act as a treatment supporter or identify treatment supporter who is accessible and acceptable to the patient to provide TB treatment
- support for adherence to treatment and monitoring of TB patients at the community level
- update the original treatment card at the PHI on a fortnightly basis and Nikshay entries in coordination with NTEP staff
5. Ensuring public health action:
The PHW visits the house of the patient within a week of TB diagnosis to:
- verify the residential address so that in case of interruption, retrieval action can be taken
- counsel the patient and family members regarding the disease, treatment and its adherence
- screening of contacts, providing TB Preventive Therapy (TPT) to all eligible
- advise patient on balanced diet, taking the food they can afford and also about nutritional support systems available for the eligible patients
- collect the bank account number of the patient or one of the household members; or facilitate getting the bank account opened, if not having one
- mobilize/refer for HIV testing
- sample collection and transportation for Dug Susceptibility Testing
6. Awareness generation/advocacy in community
PHWs generate awareness/advocacy in the community. The activities include:
- awareness on health promotion and health seeking behaviour
- awareness on symptoms of TB, good cough etiquettes, available services for screening, diagnosis and treatment of TB
- awareness on patient support and benefit schemes including Nikshay Poshan Yojana
- mobilize community, community leaders (religious leaders, school principals, women’s Self-Help Groups, etc) and Panchayati Raj Institution (PRI) members for TB sensitization activities
- identify TB survivors to volunteer for the community engagement activities
Resource
Training Modules for Programme Managers and Medical Officers, Central TB Division, MoHFW 2020
Assessment
Question
Answer 1
Answer 2
Answer 3
Answer 4
Correct answer
Correct explanation
Page id
Part of Pre-test
Part of Post-test
What is the key role of the Peripheral Healthcare Worker?
Vulnerable population mapping
Home visits, counselling and contact tracing
Awareness generation/ advocacy in community
All of the above
4
Peripheral Healthcare Workers' (PHW) role is in vulnerable population mapping, screening and referral for testing, treatment initiation, coordinating treatment support, ensuring public health action, and awareness generation/ advocacy in the community.
Yes
Yes
Role of STLS at a DMC
ContentThe Senior TB Laboratory Supervisor (STLS) is the person responsible for monitoring the day-to-day activities of all the microscopy centres and nucleic acid amplification test (NAAT) sites and is thus essential to the success of the National TB Elimination Programme (NTEP). They also ensure the quality of TB diagnostic services.
Roles of STLS at the DMC
- Program management
The STLS is responsible for ensuring that microscopy services in the district are well organized and the locations of the designated microscopy centres (DMCs) are known to all the medical officers in all peripheral health institutions (MO-PHI).
The STLS also ensures that there are:
- Uninterrupted staffing of DMCs, including coverage for laboratory technicians (LTs) that might be on leave, so that there is regular and uninterrupted availability of smear examination at the DMC.
- Uninterrupted supply of reagents and logistics required for the microscopy.
- Quality assurance and accuracy guarantee for the microscopic activities carried out.
- Regular training and continuing education of LTs.
The STLS reports to the district TB officer (DTO) in collaboration with the senior treatment supervisor (STS) regarding implementation, quality control (QC), supervision and management of laboratory supplies.
- Monitor documentation related to microscopy
STLS ensures that all documentation related to sputum smear examinations is accurate and reports of examinations are given to the treating physician promptly. This includes:
- Each LT has a TB lab register which is filled completely and accurately.
- LTs understand the importance of limiting administrative errors (for example, keeping the sputum specimens with the proper lab forms for sputum examination and slides) and accurately recording the results of sputum smear examinations.
- LTs keep examined slides for review and on-site evaluation (OSE) visit by the STLS.
- There is an accurate recording of the results of the sputum smear examination.
STLS must explain to LTs that patients are diagnosed and placed on appropriate treatment regimens based on the smear results.
- Ensure appropriate number and schedule of sputum examinations
- Presumptive TB persons should have their sputum examined the correct number of times for tubercle bacilli, at least 2 sputum samples should be examined.
- Follow-up cases should have 2 sputum samples examined and should be done according to the follow-up schedule.
- Perform laboratory QC
This is done via OSE visits. The visit includes a comprehensive assessment of the laboratory safety including infection control measures; conditions of the equipment, adequacy of supplies as well as the technical components of acid-fast bacilli (AFB) smear microscopy employing a simple “Yes” and “No” checklist.
- The STLS visits every DMC under their supervision at least once every 4 weeks, and more often if possible.
- During these visits, at least 5 positive and 5 negative slides must be re-checked by the STLS.
- Staff at the DMC is supervised, evaluated and trained during these visits.
- The STLS maintains a diary, recording the details of these field visits.
At the end of each QC visit, detailed feedback is given by the STLS for continuous internal quality improvements. The STLS also ensures that centres maintain proper storage and transport of sputum specimens, the safety of lab staff and the maintenance of microscopes.
5. Waste disposal checks: STLS ensures that contaminated material is disposed of safely to ensure infection control. Sputum containers with sputum must either be incinerated, disinfected and autoclaved, or burnt in a pit and the burnt material buried.
6. Maintain an adequate supply of all materials necessary for microscopic examination
The STLS ensures that LTs have an adequate supply of reagents, sputum containers, slides and other materials including boxes for storing slides. This includes:
- Calculating the required volume of material (slides, sputum containers, etc.) required.
- Ordering supplies during the first week of the quarter to ensure uninterrupted supply at all DMCs.
- Distribution of sputum containers to all sputum collection centres/ DMCs in the area.
- Estimating and ensuring maintenance of adequate reserve stock at DMCs.
Resources
- NTEP Training Modules 1-4 for Programme Managers & Medical Officers, 2020.
- Module for Senior Tuberculosis Laboratory Supervisors, NTEP, 1999.
- Module for Laboratory Technicians, NTEP, 2005.
Assessment
Question
Answer 1
Answer 2
Answer 3
Answer 4
Correct answer
Correct explanation
Page id
Part of Pre-test
Part of Post-test
What is the role of the STLS in the DMC?
Conduct an on-site evaluation visit.
Make sure that LTs fill the TB lab register properly.
Ensure adequate supply of lab material, like slides, at the DMC.
All of the above
4
The STLS conducts an on-site evaluation visit at the DMC, ensures proper documentation of smear results and ensures an adequate supply of lab materials at the DMC.
Yes
Yes
How often should the STLS visit the DMC?
Every 2 weeks
Every month
Every quarter
Every 6 months
2
The STLS visits every DMC under their supervision at least once every 4 weeks, and more often if possible.
Yes
Yes
Role of STS at a DMC
ContentAt a DMC, the STS primarily ensures that
1. All patient services from enrolment to outcomes for a TB patient are completed optimally. This is done by monitoring information submitted by the DMC such as referral for testing, no of people tested, no of people diagnosed with TB, initiated on treatment.
2. All the patients started on treatment are tested promptly using the appropriate follow-up testing schedule( i.e. at the end of IP and CP).
3. Maintain profile of the DMC in the Nikshay such as tagging the PHI as DMC, name of contact person and other particulars
4. Ensure data quality in the various records, both in physical and electronic records. This includes patient data, referral data and testing data.
Fullscreen