Storing slides at a DMC for QA
ContentIt is important to store slides, after reading as these will be required for Quality assurance - internal rechecking by the supervisor, and for External Quality Assurance (EQA) as per National TB Elimination Programme (NTEP) guidelines.
- Prior to storage slides need to be dried by gently placing the slide gently face down on the tissue paper or by wrapping it in the tissue paper and leaving it overnight to remove excess oil (Figure 1).

Figure 1: Using Tissue Paper to Drain Oil
- The slides are to be stored in the slide box in the same order as they are listed in the laboratory register (Figure 2).

Figure 2: Keeping the Slides in Same Order as Listed in the Laboratory Register
- One blank place must be left behind the first slide from a patient suspected with TB this allows the second slide of the same patient to be added after reading. This process will keep results consistent with the laboratory register (Figure 3).

Figure 3: Keeping Blank Spaces Between Slides for Consistency
- Always store slides in closed slide boxes away from sunlight (Figure 4).

Figure 4: Storing Slides in Closed Slide Boxes

Resources
- Manual for Sputum Smear Fluorescence Microscopy, RNTCP, MoHFW, 2007.
- Laboratory Diagnosis of Tuberculosis by Sputum Microscopy, GLI Initiative.
Kindly provide your valuable feedback on the page to the link provided HERE
False Positivity and False Negativity in Microscopy
ContentFalse result: It’s a situation where the test result is different from the truth.
Types of False Results
- False Positive: When the test result is positive but the patient does not have TB.
- False Negative: When the test result is negative but the patient has TB.
Both cases will lead to wrong categorization and incorrect treatment. The consequences of false results are shown in the figure below:
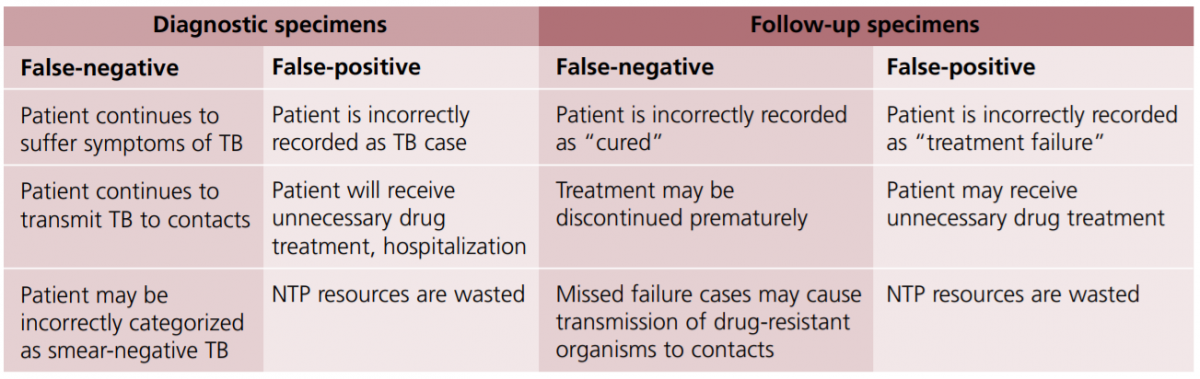
Figure: Consequences of False Results in Sputum Smear Microscopy; Source: Quality Assurance of Sputum Microscopy in DOTS Programmes.
Resources
- RNTCP Module for Laboratory Technicians, CTD, MoHFW, 2005.
- Quality Assurance of Sputum Microscopy in DOTS Programmes Regional Guidelines for Countries in the Western Pacific, WHO, 2003.
Kindly provide your valuable feedback on the page to the link provided HERE
Prevention of False Positive Results During Smear Microscopy
ContentFalse positive results can be prevented by using the following measures:
- Always use new, unscratched slides.
- Use a separate broomstick for each sample.
- Always use filtered Carbol fuchsin.
- Do not allow the Carbol fuchsin to dry during staining.
- Decolorize adequately with Sulphuric acid.
- Make sure that there are no food particles or fibers in the sputum sample.
- Never allow the oil immersion applicator to touch a slide.
- Label sputum containers, slides and laboratory forms accurately.
- Cross-check the number on the laboratory form and sputum container before recording.
- Record and report the results accurately.
Resources
Kindly provide your valuable feedback on the page to the link provided HERE
Prevention of False Negative Results During Smear Microscopy
ContentTo prevent false negative results during smear microscopy, each laboratory technician needs to:
- Make sure that the sample contains sputum, not just saliva.
- Make sure there is enough sputum (at least 2-5 ml).
- Select thick, purulent particles to make the smear.
- Prepare smears correctly - not too thick, not too thin or not too little material.
- Fix the slide for the correct length of the time, not too short or too long.
- Stain with Carbol fuchsin for full five minutes.
- Do not decolorize with Sulphuric acid too intensively.
- Examine every smear for at least five minutes and full 100 fields before recording it as negative.
- Label the sputum containers, slides and laboratory forms carefully.
- Cross check the number on the laboratory form and sputum container before recording.
- Record and report the results accurately.
Resources
Kindly provide your valuable feedback on the page to the link provided HERE
Need for Quality Testing and Control
ContentQuality Assurance (QA) is a systematic process used to determine the quality standards of TB laboratories.
Poor quality diagnosis may results in failure to diagnose TB or unnecessary treatment of non-TB case. Thus an effective QA mechanism is important for reliability of laboratory diagnosis.
QA in National Tuberculosis Elimination Programme (NTEP) consists of:
- Internal Quality Control (QC)
- External Quality Assessment (EQA)
- Continuous Quality Improvement (QI)
QC or Internal Quality Assurance is a systematic internal monitoring of working practices, technical procedures, checking equipment, new lot of reagents, smear preparation, grading etc
EQA is a process to assess laboratory performance by comparing laboratory results with a laboratory in higher tier i.e.Intermediate Reference Laboratory (IRL)/ National Reference Laboratory (NRL) through on-site evaluation, panel testing and random blinded rechecking of slides. Figure 1 depicts an eg of on-site evaluation with roles of various stakeholders and frequency of this activity.

Figure 1: On-site evaluation (RNTCP Laboratory Network Guidelines)
QI is continuous monitoring of laboratory performance, finding non compliance and taking remedial measures to prevent recurrence of problems
Resources
Assessment
Question
Answer 1
Answer 2
Answer 3
Answer 4
Correct answer
Correct explanation
Page id
Part of Pre-test
Part of Post-test
Quality Improvement mostly relies on effective on-site evaluation visits?
True
False
1
Quality Improvement mostly relies on effective on-site evaluation visits.
Yes
Yes
Measures for Quality Assurance in Microscopy
ContentSputum smear microscopy is an integral part of the National TB Elimination Programme's (NTEP’s) diagnostic services and can majorly impact the success of the programme.
It is essential to have a credible and well-established quality assurance system to assess the performance of the microscopy service.
NTEP has implemented a multi-level quality assurance network system of sputum smear microscopy in the country consisting of:
- Internal Quality Control (IQC)
- External Quality Assessment (EQA)
- Quality Improvement (QI)
- Internal Quality Control is a process of effective and systematic internal monitoring of the working practices.
- It includes technical procedures, checking instruments, quality of new batches of staining solution, smear preparation, grading, equipment infection control measures, waste management, etc.
- For the purpose of IQC, it should be ensured that all staining reagents’ dye content is mentioned on the bottle, concentrated acids are stored carefully in separate containers and stains are filtered before use.
- Further, after each new batch of reagents is made, Quality Control Positive (QCP) and Quality Control Negative (QCN) slides should be prepared by the Senior TB Lab Supervisor (STLS) for quality control.
- QCP slides should be prepared by pooling 3+ grade sputum samples, while QCN slides are to be prepared by pooling negative sputum samples with an adequate number of pus cells (≥10 pus cells/ field).
- One set of QCP-QCN slides should be stained by STLS and another set should be given to the Designated Microscopic Centre (DMC) Lab Technician (LT) along with the reagent, and the results of both should be entered in the batch register/ IQC document.
- All quality control slides should be stored for a maximum period of Four months. For the microscope, the lens should be cleaned with tissue paper after examining each slide and stored inside a microscope box at the end of the day.
- The microscope box should contain silica gel and an electric bulb of 10-15 watts for desiccation to prevent fungal growth on the lens. The silica gel should be dehydrated periodically under direct sunlight. All microscopes should be covered under the Annual Maintenance Contract (AMC) with routine preventive maintenance.
- External Quality Assessment (EQA) is a process to assess the performance of the peripheral laboratories by a more competent laboratory, like the intermediate or the national reference laboratory.
- EQA has 3 components:
1) Onsite evaluation of peripheral laboratories by the supervisors from the reference laboratory under actual working conditions, in order to review the internal quality control mechanisms in place.
2) Panel testing of the slides (unstained and stained) received from the reference laboratory by the peripheral laboratory LT to evaluate the smear microscopy process. This helps to determine whether the LT can adequately perform Acid-fast Bacilli (AFB) smear microscopy.
3) Random blinded re-checking of a small sample of routine slides (both positive and negative) in the peripheral laboratory by the reference laboratory to assess the accuracy of the reading.
- Quality Improvement (QI) is a process by which all components of smear microscopy diagnostic services are continuously monitored and carefully analysed with the aim to identify the problems and implement corrective actions (including retraining) to prevent the recurrence of the problems.
Resources
- Training Module (1-4) for Program Managers and Medical Officers, NTEP, MoHFW, 2020.
- Guidelines for Quality Assurance of Smear Microscopy for Diagnosing Tuberculosis, RNTCP Lab Network, CTD, MoHFW, India, 2005.
Assessment
Question Answer 1 Answer 2 Answer 3 Answer 4 Correct answer Correct explanation Page id Part of Pre-test Part of Post-test Which of these is systematic internal monitoring of working practices that includes technical procedures, staining solution, smear preparation, grading, equipment infection control measures and waste management? Onsite evaluative (OSE) External Quality Assessment (EQA) Internal Quality Control (IQC) Quality Improvement (QI) Internal Quality Control (IQC) Internal Quality Control (IQC) is systematic internal monitoring of working practices that includes technical procedures, staining solution, smear preparation, grading, equipment infection control measures and waste management. Yes Yes Quality control slides should be stored for a maximum period of how many months? 3 6 5 2 1 All quality control slides should be stored for a maximum period of three months.
Yes
Yes
Fullscreen
