Microscopy
ContentMicroscopy is a TB diagnostic technology that utilizes the acid-fastness property of Mycobacterium tuberculosis to visualize it under a microscope. Results of sputum smear microscopy can either be smear-negative, or smear-positive (with various grades).
Advantages:
- It is currently the most accessible and cheapest TB diagnostic test available under National TB Elimination Programme (NTEP) in India.
- It has the shortest turnaround time for diagnosis.
- It has high specificity.
Limitations:
- Low sensitivity. It becomes positive only when more than 5000 bacilli/ml of sample are present. Hence, cases would be missed in early disease, or when an inappropriate biological specimen is provided, where bacterial load in sputum is less.
- It is unable to differentiate between M. tuberculosis and Non-tuberculous Mycobacteria (NTM). This is predominantly an issue in geographies with lower burden.
There are two types of microscopies used in NTEP: Ziehl-Neelsen (ZN) Microscopy and Fluorescence Microscopy (FM). These vary in the type of stain and microscope used. FM is newer of the two types and is currently recommended for use over ZN.
Resources
Ziehl–Neelsen Microscopy
ContentIn Ziehl-Neelsen microscopy, the carbol fuchsin fuchsin stain is heated to enable the dye to penetrate and bind the waxy mycobacterial cell wall. Following acid-decolourisation, the sputum smear is counterstained with methylene blue which stains the background material, providing a contrast blue colour against which the red AFB can be seen.
On observation under a microscope with oil immersion at 100X magnification, AFB appears as red, straight, or slightly curved rods, occurring singly or in small groups, while the rest of the background, mucoid and pus cells are stained blue in colour.
The method was initially developed by Paul Ehrlich and later modified by afterwards the Franz Ziehl and Friedrich Neelsen after whom the method is named.
Video fileResources
- Laboratory Diagnosis by Sputum Smear Microscopy - The Handbook, GLI, 2013.
- Module for Laboratory Technicians, CTD, 2005.
Assessment
Question Answer 1 Answer 2 Answer 3 Answer 4 Correct answer Correct explanation Page id Part of Pre-test Part of Post-test In ZN microcopy what is the stain used Auramine Carbol fucshin Methylene blue Potassium Permanganate 2 The stain used in ZN microscopy is Carbol Fucshin responsible for the reddish color of the bacteria. Yes Yes Fluorescence Microscopy Using LED Microscope
Content
Fluorescence Microscopy is a newer and better type of microscopy where the TB bacteria are stained using a fluorescent dye using the property of Acid Fastness. The dye will fluoresce when illuminated by UV light. When the UV light source is an LED Lamp, it is called LED fluorescent microscopy.
Here the bacilli appear as slender bright yellow fluorescent rods, standing out clearly against a dark background, as can be seen in the figure below.
Figure: AFB as seen under an LED Fluorescence Microscope:
Principle of Fluorescence Microscopy
- Cell walls of Acid-fast Bacilli (AFB) is made up of Mycolic Acid. The mycolic acid creates a waxy layer, making the cell wall impermeable to acids and alkalis.

- The primary stain, a fluorescent dye called Auramine-O, binds to the cell wall of the bacilli.

- Intense decolorization by acid alcohol does not release the primary stain. Thus, the AFB retain the colour of the primary stain, while other bacteria lose the stain
- The counterstain, Potassium Permanganate provides a contrasting background and is useful to quench background fluorescence.
Advantages of LED-FM:
- Fluorescence LED microscopy is more sensitive (10%) than conventional ZN microscopy.
- It may be placed in existing DMCs and does not require any additional infrastructure.
- Examination of fluorochrome-stained smears takes less time.
Video fileResources
- Cell walls of Acid-fast Bacilli (AFB) is made up of Mycolic Acid. The mycolic acid creates a waxy layer, making the cell wall impermeable to acids and alkalis.
Cartridge Based Nucleic Acid Amplification Test [CBNAAT]
ContentCartridge Based Nucleic Acid Amplification Test (CBNAAT) is a rapid molecular diagnostic test. It is used for diagnosis of Tuberculosis (TB) and Rif-resistant Tuberculosis (RR-TB) in NTEP. Results are obtained from unprocessed sputum samples in about 2hours which helps in early detection and treatment of TB patients.
India has vast number of CBNAAT laboratories which are utilized for TB/RR-TB detection and Universal Drug Susceptibility Testing (UDST) under the National TB Elimination Program (NTEP).

Figure: CBNAAT Cartridge and Machine in Use (Image courtesy: USAID supported Challenge TB Project)
The CB-NAAT system detects DNA sequences specific for Mycobacterium tuberculosis complex and rifampicin resistance by Polymerase Chain Reaction (PCR). It concentrates Mycobacterium tuberculosis bacilli from sputum samples, isolates genomic material from the captured bacteria by sonication and subsequently amplifies the genomic DNA by PCR. The process identifies clinically relevant rifampicin resistance-inducing mutations in the RNA polymerase beta (rpoB) gene in the Mycobacterium tuberculosis genome in a real-time format using fluorescent probes called molecular beacons.
Video fileVideo: Cartridge-Based Nucleic Acid Amplification Test [CBNAAT] - GeneXpert Technology
Resources
- Training Module (1-4) for Program Managers and Medical Officers, NTEP, MoHFW, 2020.
- India TB Report 2021, National TB Elimination Program (NTEP), MoHFW, 2021.
Assessment Questions
Question
Answer 1
Answer 2
Answer3
Answer 4
Correct Answer
Correct explanation
Part of pre-test
Part of post-test
Under NTEP, CBNAAT is offered upfront for which of these categories?
PLHIV
Paediatric presumptive TB
Presumptive DR-TB
All of the above
4
Under NTEP, CBNAAT is recommended upfront for People living with HIV, Paediatric Presumptive TB patients, Presumptive DR-TB patients and patients notified from the Private sector.
Yes
Yes
CBNAAT requires the processing of sputum samples before testing
True
False
2
Results are obtained from unprocessed sputum samples in about 2hours from a CBNAAT machine
Yes
Yes
Truenat
ContentTruenat is an indigenous rapid molecular test platform that is currently under use in NTEP for diagnosis of TB and Rif Resistance. It is a platform utilising real-time Polymerase Chain Reaction (PCR) technology built into micro-PCR chips.
Testing on Truenat involves three components:
- Workstation (consisting of 2 devices)
- Trueprep AUTO Universal Cartridge-based Sample Prep Device for the automated extraction and purification of DNA
- Truelab Real-time micro PCR Analyzer for performing real-time PCR. It is available as 1 (Uno), 2 (Duo) or 4 (Quattro) chip ports.
- Cartridge and Chip
- Reagent kits (Sample Pre-treatment and Prep kits)

Figure: Truenat Source: MolBio Products.
Test results for MTB detection and Rif Resistance has a turn around time of 1-2 hours. Depending on the micro-PCR chips used various tests can be performed using Truenat. Truenat MTB micro-PCR chips detect Mycobacterium tuberculosis bacteria for TB diagnosis. Truenat MTB RIF micro-PCR chip is used as a reflex test to detect resistance to Rifampicin (RIF), the first-line drug for TB treatment
Truenat has many advantages. Truenat is designed to be mobile and is battery operated (~8 hours on full charge). It can be deployed in peripheral laboratories and microscopy centres with minimal or no added facilities and hence it is more point-of-care. Biosafety requirements are similar to smear microscopy. However, it is multi staged and partially automated, requiring the presence of a Lab Technician through out the test.
Resources
- Truenat MTB Kit Insert.
- Trueprep AUTO Universal Cartridge-based Sample Prep Device.
- Practical Guide to Implementation of Truenat Tests for the Detection of TB and Rifampicin-resistance, 2021.
Assessment
Question
Answer 1
Answer 2
Answer 3
Answer 4
Correct answer
Correct explanation
Page id
Part of Pre-test
Part of Post-test
Truenat is used in NTEP for: MTB detection Rif Resistance Detection INH resistance Detection MTB and Rif Resistance Detection 4 Truenat is used for MTB and Rif Resistance detection in NTEP Yes Yes The Truelab Analyzer is available in how many chip ports?
2 (Duo)
1 (Uno), 2 (Duo) and 4 (Quattro)
1 (Uno)
4 (Quattro)
2
The Truelab Analyzer is available as 1 (Uno), 2 (Duo) and 4 (Quattro) chip ports.
Yes Yes - Workstation (consisting of 2 devices)
Line Probe Assay [LPA]
ContentLine Probe Assay (LPA) is a rapid molecular test available at centralised laboratories.
The assay is based on Polymerase Chain Reaction (PCR) that can simultaneously detect Mycobacterium tuberculosis complex as well as drug sensitivity to anti-TB drugs.

Figure 1: The GenoType MTBDRplus Molecular LPA Procedure; Source: Molecular Detection of Drug-resistant Tuberculosis by Line Probe Assay.
Advantages of LPA
- Rapid molecular test. (Turnaround time: 3-5 days)
- Highly sensitive and specific.
- Performed directly from sputum smear-positive specimens and on isolates of M. tuberculosis complex grown from smear-negative and smear-positive specimens.
- Detects multiple gene mutations in anti-TB drugs.
- First-line LPA detects mutations to rifampicin and isoniazid
- Second-line LPA detects mutations to fluoroquinolones and aminoglycosides.
- Suitable for low and high-throughput labs.
Disadvantages of LPA
- Cannot be used as a point-of-care test.
- Requires appropriate laboratory infrastructure, equipment and biosafety precautions.
- Different rooms (DNA extraction, pre-amplification, amplification, post-amplification/ hybridization) are required to perform different steps (Figure 2).
- Requires trained manpower to perform tests and interpret test results.
- Stringent internal quality control is required to prevent contamination.

Figure 2: Amplification (A) and Post-amplification Laboratory (B) for LPA; Source: Molecular Detection of Drug-resistant Tuberculosis by Line Probe Assay.
Resources
- Guidelines for PMDT in India, 2021.
- Molecular Detection of Drug-resistant Tuberculosis by Line Probe Assay.
Assessment
Question Answer 1 Answer 2 Answer 3 Answer 4 Correct answer Correct explanation Page id Part of Pre-test Part of Post-test LPA can be used as a point-of-care test. True False 2 LPA cannot be used as a point-of-care test. Yes Yes Solid and Liquid Culture in TB
ContentCulturing TB Bacilli is well known and historic method for detection/ confirmation of Tuberculosis. It is a highly sensitive and specific phenotypic test; it can detect even a few viable bacilli in the sample (Upto 10 Colony Forming Units- CFUs). TB bacilli multiply in the culture and form colonies of TB bacilli which can are easily be identified.
Based on the growth media used Culture is divided in to two types, Solid and Liquid Culture methods. Types Culture:
- Solid Culture on Lowenstein Jensen media : Historic gold standard culture test. Results take usually upto 2 months (60 days).
- Modern Liquid culture systems: (e.g. BACTEC MGIT 960, BacT Alert or Versatrek etc.) Results take usually up to 42 days.
Uses
- Solid culture is the gold standard diagnostic test for TB. But it is not used for the purpose of TB diagnosis due to the long turn around time of 2 months. It is largely used for research purposes where it is used as the baseline test on which the sensitivity and specificity of other tests are calculated.
- Liquid Culture is being used for follow-up monitoring of patients on drug resistant TB treatment to detect treatment failure. Liquid culture is also used for long term follow up patients who have successfully completed treatment to detect recurrence.
- Liquid culture is used as a previous step to grow bacilli and obtain isolates prior to Drug Susceptibility Testing.
- Liquid cultures are also used in TB prevalence surveys for its high sensitivity and specificity
Resources
Kindly provide your valuable feedback on the page to the link provided HERE
Culture Drug Susceptibility Testing [CDST]
ContentCulture Drug Susceptibility Testing (CDST) is a growth-based phenotypic method used to check the susceptibility of Mycobacterium tuberculosis strains to various first and second line anti-TB drugs. Mycobacterial resistance to a particular drug is identified if there is growth observed in culture in presence of that drug.
In NTEP CDST is the standard method to detect resistance in samples of patients who have tested positive on followup. While CDST is possible on both Solid and Liquid culture, currently, the NTEP utilizes only liquid culture as a method for DST, due to faster Turn around times.
CDST testing services are available under NTEP in designated, specialized laboratories called CDST Labs both in public and private sector. Currently there are 80 such laboratories (60 certified for First Line and 49 for Second line drugs). Such designated laboratories are subject to regular external quality assessment, often by the National Reference Laboratory at that region.
Quality assured DST to R, H, Z, Mfx, Lfx, Lzd, Am, Km and Cm are available across the country.
Resources
Monitoring of Treatment by Smear and Culture
ContentSmear microscopy and culture play an important role in monitoring the response to TB treatment. In NTEP, smear microscopy and culture has been prescribed to monitor the treatment response and at specific time points based on different treatment regimens. The programme also explains the actions that need to be taken based on the follow-up results.
Monitoring of tuberculosis treatment is shown in the table below.
Table: Monitoring of TB treatment by smear and culture
TYPE OF TREATMENT
MONITORING BY SMEAR
MONITORING BY CULTURE
ACTION TO BE TAKEN FOR FOLLOW UP
Drug Sensitive Pulmonary TB
One sputum smear examination at time of completion of the intensive phase (IP) of treatment and at the end of treatment
-
If sputum is positive anytime during treatment, then drug susceptibility testing (DST) is to be done. Further treatment is guided by the DST pattern.
H mono/poly DR-TB
Monthly from month 3 onwards till the end of treatment. Conduct smear microscopy within 7 days, if the smear at month 4 or later is positive to rapidly ascertain bacteriological conversion/reversion
At the end of month 3, end of treatment (month 6 and/or 9 if applicable)
If the culture results of month 3 are positive, the culture is repeated to rapidly ascertain bacteriological conversion/ reversion. If the repeat specimen is culture negative, culture is done at the end of treatment
Shorter oral Bedaquiline-containing MDR/RR-TB regimen
Monthly from 3rd month onwards till end of IP monthly in extended IP only if previous month is smear positive ( S+ve). Conduct SM within 7 days, if the smear at 6 months is positive to rapidly ascertain bacteriological conversion/reversion.
At the end of month 3, end of month 6 and/or end of treatment.
If the culture results of month 6 is positive, collect one repeat sample immediately to rapidly ascertain the bacteriological conversion/reversion. If the repeat sample is culture negative, then conduct an end of treatment specimen collection.
Longer Oral M/XDR-TB Regimen
Examine sputum smear within 7 days and every time C&DST is done.
Monthly from month 3 onwards to end of 6 months or 7 or 8 if the previous month’s culture is positive; Quarterly month 6 or 7 or 8 onwards based on previous month’s culture results
If the smear/culture results of month 6 or any of the quarterly culture is positive, repeat culture is done to ascertain bacteriological conversion/reversion and if the repeat specimen is culture negative, then culture is done the subsequent quarter or end of treatment
Resource
Kindly provide your valuable feedback on the page to the link provided HERE
Culture Conversion
ContentSputum culture conversion - The transition in sputum culture results from a positive sample growing Mycobacterium tuberculosis to two consecutive negative cultures separated by at least 30 days.
Time to culture conversion is the interval between treatment initiation to date of specimen collection of the first (of the two) negative culture.
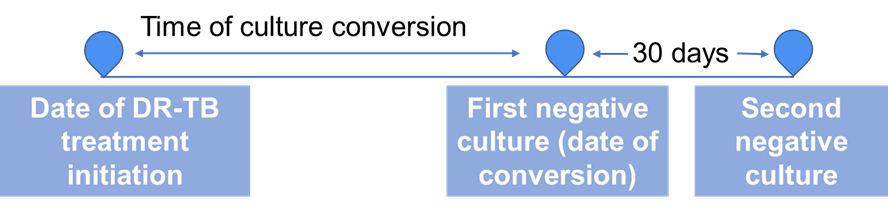
Culture conversion is important indicator to assess the effectiveness of a treatment regimen for Drug-resistant Tuberculosis (DR-TB).
All DR-TB cases are followed up periodically for culture after initiation of the treatment. Based on the culture results and other parameters, the course of treatment is decided.
Resources
- Time to Culture Conversion and Regimen Composition in Multidrug-resistant Tuberculosis Treatment; Tierney D. Harvard Library
- Guidelines for Programmatic Management of Drug-resistant Tuberculosis in India, 2021.
Kindly provide your valuable feedback on the page to the link provided HERE
Fullscreen