Visualization of CBNAAT Test Results
ContentThe test results of the Cartridge-based Nucleic Acid Amplification Test (CBNAAT) assay are displayed in the ‘View Results’ window, of the CBNAAT software.
For visualizing the results after the test is completed:
- In the CBNAAT Dx System window, click View Results on the menu bar.
- The View Results window appears.
- To select a test result, click View Test.
- ‘Select Test to Be Viewed’ - dialog box appears.
- Select the test of interest.
- Click OK.
Note: The selected test result appears in the ‘View Results’ window. A result will be displayed in PDF format.
To generate a PDF report
- To generate a report in PDF format, click on “Report” and then select the result you want.
- The PDF report will be generated.
Interpretation of Test Results
- The ‘View Results’ window displays information about the test, such as sample ID and run-time on the left-hand side panel.
- The interpretation of the result is in the center, and the real-time PCR amplification curves are displayed at the bottom.
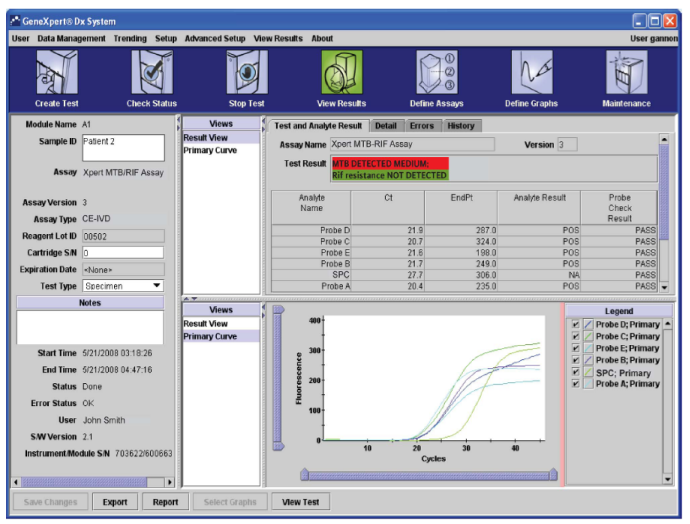
Figure: User 'View Results' Window showing the information about test, interpretation of results and real-time PCR curves
Resources
Kindly provide your valuable feedback on the page to the link provided HERE
CBNAAT Results Interpretation
ContentOn completion of a test run, the Cartridge-based Nucleic Acid Amplification Test (CBNAAT) gives the following results:
- MTB DETECTED; Rif Resistance DETECTED
- MTB DETECTED; Rif Resistance NOT DETECTED
- MTB DETECTED; Rif Resistance INDETERMINATE
- MTB NOT DETECTED
- Error
- Invalid
- No result
Conclusive results include: MTB NOT DETECTED, MTB DETECTED with Rif Resistance/ without Rif Resistance.
Non-conclusive results include: MTB Detected, Rif Resistance Indeterminate, Errors, Invalid and No Result - the test has to be repeated in these cases.
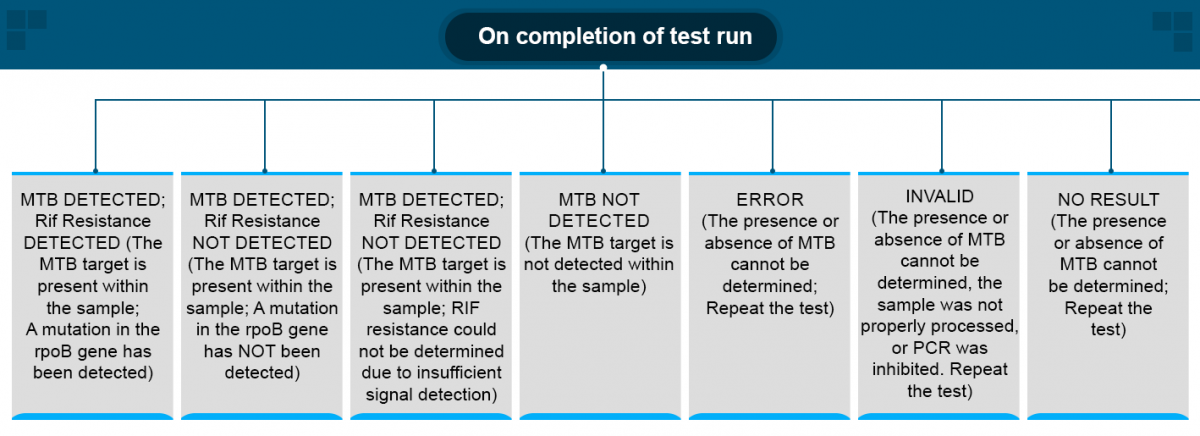
Figure: CBNAAT Result Algorithm
Video fileVideo : CBNAAT Results Interpretation
Resources
CBNAAT Assay Limitations
ContentThe Cartridge-based Nucleic Acid Amplification Test (CBNAAT) test has some limitations such as:
- Bacterial load below the Limit of Detection (LOD ~ 130 CFU/ml) may result in a false-negative result.
- Patients on an anti-TB regimen can still have positive results due to killed bacilli in the specimen and hence cannot be used for follow-up.
- A positive test result does not necessarily indicate the presence of viable organisms. It is, however, presumptive for the presence of Mycobacterium tuberculosis (MTB) and Rifampicin (Rif) resistance.
- MTB detection is dependent on the number of organisms present in the sample. Quality specimen collection and timely processing of the sample will minimize the errors.
- Test results might be affected by anti-TB medication. Therefore, therapeutic success or failure cannot be assessed using this test, because DNA might persist following antimicrobial therapy.
- Mutations or polymorphisms in primer or probe binding regions may affect the detection of new or unknown MDR or Rif-resistant strains, resulting in a false-negative result.
- Any modification in sample processing may alter the performance of the test.
- Results should be interpreted in conjunction with clinical data available to the clinician.
Video fileVideo:
CBNAAT Assay Limitations
Resources
Retest Procedure for CBNAAT
ContentThe assay needs to be repeated by using a new cartridge if one of the following test results occur:
INVALID: An INVALID result indicates that Sample Processing Control (SPC) failed. The sample was not properly processed, or Polymerase Chain Reaction (PCR) was inhibited.
ERROR: An ERROR indicates that Probe Check Control (PCC) failed, and the assay was aborted possibly due to the reaction chamber being filled improperly, a reagent probe integrity, syringe pressure issues, or failure of the CBNAAT module.
NO RESULT: A NO RESULT indicates that insufficient data were collected. For example, the operator stopped a test that was in progress.
RIF Indeterminate: RIF indeterminate result indicates that the sample has a less bacterial load.
Program guidelines recommend obtaining a second specimen to confirm rifampicin resistance in these scenarios.
How to Perform the Retest?
- Leftover sputum or fresh sputum or reconstituted sediment:
Treat it with a new Sample Reagent (SR) and load it into the new CBNAAT cartridge.
- Sufficient leftover SR-treated sample:
Use within 4 hours of initial addition of SR to the sample - Load into the new CBNAAT cartridge.
Do not use SR-treated sample if it is more than 4 hours old - Over-treatment may lead to false-positive test results.
Always use a new cartridge!
Video fileVideo : Retest Protocol for CBNAAT
Resources
Fullscreen